408271
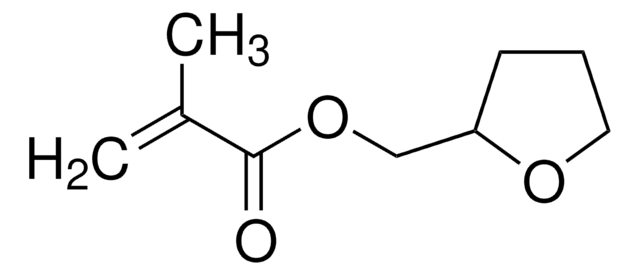
Tetrahydrofurfuryl acrylate
contains 500 ppm hydroquinone as inhibitor, 500 ppm monomethyl ether hydroquinone as inhibitor
Synonym(s):
THFA
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C8H12O3
CAS Number:
Molecular Weight:
156.18
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
form
liquid
Quality Level
contains
500 ppm hydroquinone as inhibitor
500 ppm monomethyl ether hydroquinone as inhibitor
refractive index
n20/D 1.46 (lit.)
bp
87 °C/9 mmHg (lit.)
density
1.064 g/mL at 25 °C (lit.)
SMILES string
C=CC(=O)OCC1CCCO1
InChI
1S/C8H12O3/c1-2-8(9)11-6-7-4-3-5-10-7/h2,7H,1,3-6H2
InChI key
YNXCGLKMOXLBOD-UHFFFAOYSA-N
Related Categories
Application
Tetrahydrofurfuryl acrylate may be used as an acrylic matrix for silver nanoparticles/polymer nanocomposites . It can form copolymers with butadiene. Prior to transfer printing, cellulosic and proteinic fibers are grafted with THFA.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Some new butadiene copolymers.
Marvel CS, et al.
Journal of Polymer Science, 8(6), 599-605 (1952)
Transfer printing of cellulosic and proteinic fabrics.
El-Molla MM, et al.
Advances in Polymer Technology, 20(4), 296-304 (2001)
Fan Xie et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 22(5), 455-460 (2021-01-17)
A chiral adduct formed between a chiral carboxylic acid, tetrahydro-2-furoic acid (THFA), and a chiral ester, propylene oxide (PO), was investigated using rotational spectroscopy and DFT calculations. Isolated THFA exists dominantly as three different conformers: I, II, and III in
David A Korasick et al.
Biophysical journal, 114(12), 2833-2843 (2018-06-21)
Homooligomerization of proline utilization A (PutA) bifunctional flavoenzymes is intimately tied to catalytic function and substrate channeling. PutA from Bradyrhizobium japonicum (BjPutA) is unique among PutAs in that it forms a tetramer in solution. Curiously, a dimeric BjPutA hot spot
Masao Doi et al.
Nature communications, 2, 327-327 (2011-05-26)
Synchronous oscillations of thousands of cellular clocks in the suprachiasmatic nucleus (SCN), the circadian centre, are coordinated by precisely timed cell-cell communication, the principle of which is largely unknown. Here we show that the amount of RGS16 (regulator of G
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service