381551
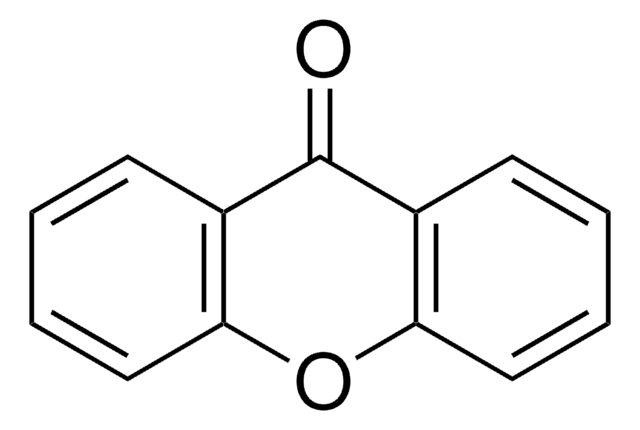
7-Isopropoxy-3-phenyl-4H-1-benzopyran-4-one
97%
Synonym(s):
Ipriflavone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C18H16O3
CAS Number:
Molecular Weight:
280.32
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
116-120 °C (lit.)
functional group
ketone
phenyl
SMILES string
CC(C)Oc1ccc2c(OC=C(C2=O)c3ccccc3)c1
InChI
1S/C18H16O3/c1-12(2)21-14-8-9-15-17(10-14)20-11-16(18(15)19)13-6-4-3-5-7-13/h3-12H,1-2H3
InChI key
SFBODOKJTYAUCM-UHFFFAOYSA-N
Gene Information
rat ... Alpl(25586)
Looking for similar products? Visit Product Comparison Guide
General description
7-Isopropoxy-3-phenyl-4H-1-benzopyran-4-one (Ipriflavone), a synthetic flavonoid, is reported to stimulate the activity of osteoblasts. It is reported to promote the deposition of calcium and the formation of mineralized nodules by newborn rat calvarial osteoblast-like (ROB) cells as well as the activity of alkaline phosphatase. Ipriflavone, an isoflavone derivative, is a new drug used to decrease bone loss in osteoporosis.
Application
7-Isopropoxy-3-phenyl-4H-1-benzopyran-4-one (Ipriflavone) has been used as a model drug in a study to functionalize the mesoporous bioactive glasses (MBG). Study suggested that since ipriflavone is a hydrophobic anti-osteoporotic drug, it easily attaches to the surface of MBG and results in long-term drug delivery.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Dae Y Lee et al.
European journal of pharmaceutical sciences : official journal of the European Federation for Pharmaceutical Sciences, 38(5), 465-471 (2009-09-19)
Ipriflavone was reported to be primarily metabolized via hepatic cytochrome P450 (CYP) 1A1/2 and 2C11 in male Sprague-Dawley rats. The protein expression and/or mRNA levels of hepatic CYP1A subfamily and 2C11 was reported to be increased and decreased, respectively, in
J Yao et al.
Poultry science, 86(3), 503-507 (2007-02-14)
The effects of ipriflavone on caged layer bone metabolism were examined in vitro and in vivo. Ipriflavone at 10(-8) M stimulated the activity of osteoblasts cultured from embryonic chick calvariae, and 10(-9) to 10(-7) M inhibited osteoclasts from chick tibias
Y Moon et al.
Xenobiotica; the fate of foreign compounds in biological systems, 37(3), 246-259 (2007-07-13)
Ipriflavone, a synthetic flavonoid for the prevention and treatment of osteoporosis, has been reported to be extensively metabolized in man to seven metabolites (M1-M7). This study was performed to characterize the human liver cytochrome P450s (CYP) responsible for the metabolism
Luciano Belcavello et al.
Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 50(3-4), 996-1000 (2011-12-28)
Ipriflavone (7-isopropoxy-isoflavone) is a semisynthetic isoflavone derivative from daidzein and prescribed to prevent and treat osteoporosis in postmenopausal women. In the present study, ipriflavone was investigated with regard to their cytotoxic and mutagenic effects using the micronucleus assay (MN) in
Xiao-Wu Xu et al.
Cellular and molecular neurobiology, 28(6), 875-886 (2008-03-04)
The present study was undertaken to evaluate whether estrogen deprivation might lead to mitochondrial alteration of hippocampal neurons of ovariectomized (OVX) rats, and to evaluate the protective effect of estrogen and phytoestrogen on the mitochondrial alteration. First, OVX rats were
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service