230707
n-Butyllithium solution
2.5 M in hexanes
Synonym(s):
n-BuLi, Butyl lithium, Butyllithium solution, Lithium-1-butanide
About This Item
Recommended Products
form
liquid
Quality Level
concentration
2.5 M in hexanes
density
0.693 g/mL at 25 °C
storage temp.
2-8°C
SMILES string
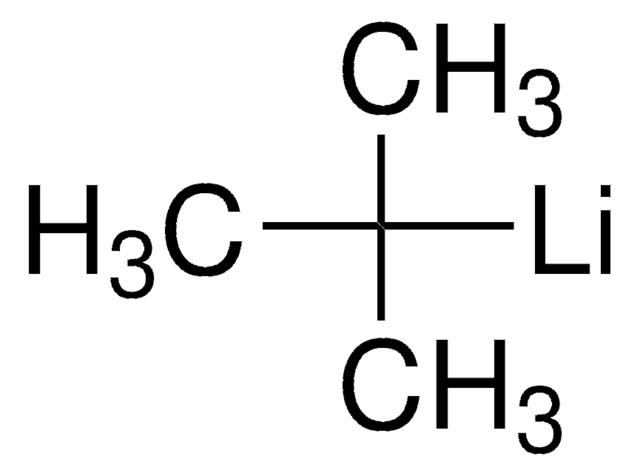
[Li]CCCC
InChI
1S/C4H9.Li/c1-3-4-2;/h1,3-4H2,2H3;
InChI key
MZRVEZGGRBJDDB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Application
The product is also used in the following reactions:
- Anionic rearrangement reactions
- Metal-halogen interchange and transmetalation reactions
- Elimination reactions
- [1,2]- and [1,4]-Wittig rearrangement reaction
- Anionic homo-Fries rearrangement reaction
- Asymmetric carbolithiation
Packaging
Legal Information
recommended
related product
Signal Word
Danger
Hazard Statements
Hazard Classifications
Aquatic Chronic 2 - Asp. Tox. 1 - Eye Dam. 1 - Flam. Liq. 2 - Pyr. Liq. 1 - Repr. 2 - Skin Corr. 1B - STOT SE 3 - Water-react 1
Target Organs
Central nervous system
Supplementary Hazards
Storage Class Code
4.2 - Pyrophoric and self-heating hazardous materials
WGK
WGK 3
Flash Point(F)
-7.6 °F - closed cup
Flash Point(C)
-22 °C - closed cup
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Transformative reagents enable selective conversions within molecules containing sensitive functionalities under mild reactions.
Transformative reagents enable selective conversions within molecules containing sensitive functionalities under mild reactions.
Transformative reagents enable selective conversions within molecules containing sensitive functionalities under mild reactions.
Transformative reagents enable selective conversions within molecules containing sensitive functionalities under mild reactions.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service