5.04908
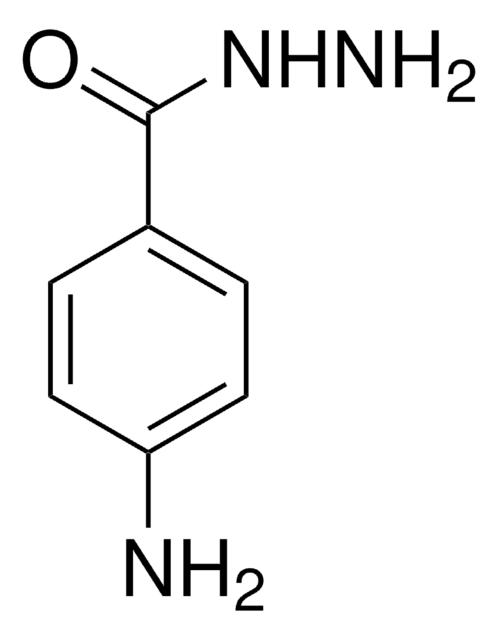
Myeloperoxidase Inhibitor-II
Synonym(s):
Myeloperoxidase Inhibitor-II, 4-(5-Fluoro-1H-indol-3-yl)butanamide, MPO Inhibitor II
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C12H13FN2O
CAS Number:
Molecular Weight:
220.24
UNSPSC Code:
12352200
NACRES:
NA.77
Recommended Products
Assay
≥95% (HPLC)
Quality Level
form
powder
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
protect from light
color
light beige
solubility
DMSO: 100 mg/mL
storage temp.
2-8°C
General description
A 5-fluorotryptamine derivative that inhibitis the MPO-catalyzed Cl-/chloride-to-OCl-/hypochlorite oxidation in the presence of H2O2 (IC50 = 18 nM by taurine/2-aminoethan-sulfonic acid chlorination assay) as well as MPO-mediated LDL ApoB-100 (ApoB100; apolipoprotein B-100) oxidation (IC50 = 18 nM by ELISA) via direct affinity binding at the myeloperoxidase active site through ϖ-ϖ interaction between its indole and the the enzyme′s heme pyrrole ring D, while exhibiting much reduced affinity toward serotonin reuptake transporter/SERT (Ki = 631 nM). Kinetic studies reveal that the inhibitor acts as a good one-electron donor for the intermediate Compound I "Fe(IV)=O...Por•+", facilitating the formation (k3 = 1,6 x 107 M-1 s-1) of Compound II (Fe(IV)-OH...Por) and thereby preventing the reduction of Compound I directly to the resting state "Fe(IV)...Por" via the "halogenation cycle", while, on the other hand, behaves as a poor electron donor for Compound II, thereby preventing the completion of the "peroxidase cycle" by stalling the further reduction of Compound II to the resting state (k4 = 92 M-1 s-1). Toxicology study using Wistar Han male rats reports no signs of adverse effects post single 10 mg/kg i.p. dosage, while signs of adverse effects are observed within the 24 h, but not after, post a higher i.p. dosage at 100 mg/kg.
A 5-fluorotryptamine derivative that inhibits myeloperoxidase/MPO activity (IC50 = 18 nM) via direct affinity interaction with enzyme active site heme pyrrole ring D, while exhibiting much reduced affinity toward serotonin reuptake transporter/SERT (Ki = 631 nM). Kinetic studies reveal that the inhibitor acts as a good one-electron donor for the intermediate Compound I "Fe(IV)=O...Por•+", facilitating the formation of Compound II (Fe(IV)-OH...Por) and thereby preventing the reduction of Compound I directly to the resting state "Fe(IV)...Por" via the "halogenation cycle", while, on the other hand, behaves as a poor electron donor for Compound II, thereby preventing the completion of the "peroxidase cycle" by stalling the further reduction of Compound II to the resting state.
Please note that the molecular weight for this compound is batch-specific due to variable water content.
Please note that the molecular weight for this compound is batch-specific due to variable water content.
Biochem/physiol Actions
Cell permeable: yes
Primary Target
myeloperoxidase
myeloperoxidase
Reversible: yes
Packaging
Packaged under inert gas
Warning
Toxicity: Standard Handling (A)
Reconstitution
Following reconstitution, aliquot and freeze (-20°C). Stock solutions are stable for up to 6 months at -20°C.
Use only fresh DMSO for reconstitution.
Other Notes
Soubhye, J., et al. 2013. J. Med. Chem.56, 3943.
Soubhye, J., et al. 2010. J. Med. Chem.53, 8747.
Soubhye, J., et al. 2010. J. Med. Chem.53, 8747.
Legal Information
CALBIOCHEM is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service