All Photos(1)
About This Item
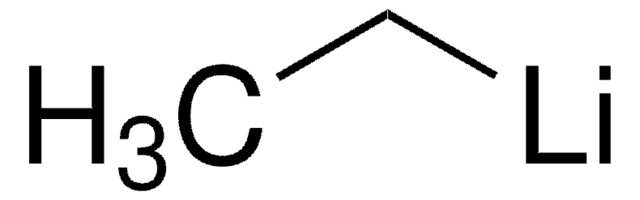
Empirical Formula (Hill Notation):
C6H7N5
CAS Number:
Molecular Weight:
149.15
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
mp
300-305 °C (lit.)
SMILES string
Cn1cnc2c(N)ncnc12
InChI
1S/C6H7N5/c1-11-3-10-4-5(7)8-2-9-6(4)11/h2-3H,1H3,(H2,7,8,9)
InChI key
WRXCXOUDSPTXNX-UHFFFAOYSA-N
Gene Information
rat ... Adora1(29290) , Adora2a(25369)
Related Categories
General description
9-Methyladenine is a derivative of adenine. It belongs to the monoclinic crystal system and P21/c space group.
Application
9-Methyladenine may be used in the preparation of N6-benzoyl-9-methyladenine.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Gydo C P van Zundert et al.
Chemphyschem : a European journal of chemical physics and physical chemistry, 12(10), 1921-1927 (2011-05-28)
IR spectroscopy is employed to study isolated adenine and its derivative 9-methyladenine in both their neutral and protonated forms. The IR spectra of neutral adenine and 9-methyladenine are measured in a molecular beam expansion via IR-UV ion-dip spectroscopy in the
Chris T Middleton et al.
The journal of physical chemistry. A, 111(42), 10460-10467 (2007-10-04)
Vibrational cooling by 9-methyladenine was studied in a series of solvents by femtosecond transient absorption spectroscopy. Signals at UV and near-UV probe wavelengths were assigned to hot ground state population created by ultrafast internal conversion following electronic excitation by a
Excited state spectroscopy and dynamics of isolated adenine and 9-methyladenine.
Luhrs DC, et al.
Physical Chemistry Chemical Physics, 3(10), 1827-1831 (2001)
Sulayman A Oladepo et al.
The journal of physical chemistry. B, 115(19), 6149-6156 (2011-04-23)
The photophysics and photochemistry of nucleobases are the factors governing the photostability of DNA and RNA, since they are the UV chromophores in nucleic acids. Because the formation of photoproducts involves structural changes in the excited electronic state, we study
Susana Ibáñez et al.
Inorganic chemistry, 50(20), 10439-10447 (2011-09-24)
The synthesis and characterization of three heteronuclear Pt(2)Pd(2) (4, 5) and PtPd(2) (6) complexes of the model nucleobase 9-methyladenine (9-MeA) is reported. The compounds were prepared by reacting [Pt(NH(3))(3)(9-MeA-N7)](ClO(4))(2) (1) with [Pd(en)(H(2)O)(2)](ClO(4))(2) at different ratios r between Pt and Pd
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service