All Photos(1)
About This Item
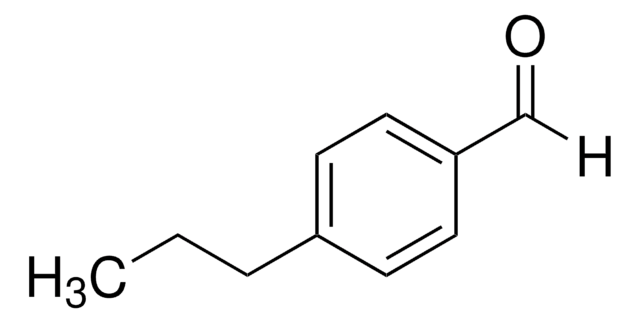
Linear Formula:
CH3(CH2)3C6H4CHO
CAS Number:
Molecular Weight:
162.23
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
90%
refractive index
n20/D 1.522 (lit.)
bp
250-263 °C (lit.)
density
0.968 g/mL at 25 °C (lit.)
SMILES string
CCCCc1ccc(C=O)cc1
InChI
1S/C11H14O/c1-2-3-4-10-5-7-11(9-12)8-6-10/h5-9H,2-4H2,1H3
InChI key
ARIREUPIXAKDAY-UHFFFAOYSA-N
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Central linkage influence upon mesomorphic and electrooptical behavior of diaryl nematics. General proton magnetic resonance method employing a lanthanide shift reagent for analysis of isomeric azoxybenzenes.
Rondeau RE, et al.
Journal of the American Chemical Society, 94(4), 1096-1102 (1972)
Francis Giraud et al.
Journal of enzyme inhibition and medicinal chemistry, 24(5), 1067-1075 (2009-06-27)
A new series of 1-benzyl-3-(imidazol-1-ylmethyl)indoles were synthesized according to a previous 3D-QSAR predictive model and assayed for their antiparasitic activity upon Leishmania mexicana promastigotes. The biological results obtained for these twelve molecules showed an IC(50) ranging from 2.3 to 32
Chao-Bin Xue et al.
Bioorganic & medicinal chemistry, 15(5), 2006-2015 (2007-01-30)
Phenoloxidase (PO), also known as tyrosinase, is a key enzyme in insect development, responsible for catalyzing the hydroxylation of tyrosine into o-diphenols and the oxidation of o-diphenols into o-quinones. Inhibition of PO may provide a basis for novel environmentally friendly
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service