5.04314
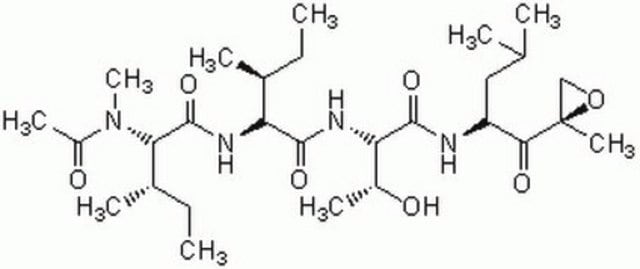
Bortezomib
≥98% (LC/MS), solid, 20S proteasome inhibitor, Calbiochem®
Sinónimos:
Bortezomib, (R)-3-Methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxamido)propanamido)butylboronic acid, BTZ, LDP-341, LDP341, MG341, MLN-341, MLN341, PS341, Proteasome Inhibitor XXII, PS-341, MG-341, Pyz-Phe-boroLeu, BTZ, LDP-341, LDP341, MG341, MLN-341, MLN341, PS341, Proteasome Inhibitor XXII, PS-341, MG-341, Pyz-Phe-boroLeu, (R)-3-Methyl-1-((S)-3-phenyl-2-(pyrazine-2-carboxamido)propanamido)butylboronic acid
About This Item
Productos recomendados
Nombre del producto
Bortezomib,
assay
≥98% (LC/MS)
Quality Level
form
solid
manufacturer/tradename
Calbiochem®
storage condition
OK to freeze
desiccated (hygroscopic)
protect from light
color
off-white
solubility
DMSO: 100 mg/mL
ethanol: 2 mg/mL (with sonication)
storage temp.
−20°C
SMILES string
B(O)(O)[C@@H](NC(=O)[C@@H](NC(=O)c2nccnc2)Cc1ccccc1)CC(C)C
InChI
1S/C19H25BN4O4/c1-13(2)10-17(20(27)28)24-18(25)15(11-14-6-4-3-5-7-14)23-19(26)16-12-21-8-9-22-16/h3-9,12-13,15,17,27-28H,10-11H2,1-2H3,(H,23,26)(H,24,25)/t15-,17-/m0/s1
InChI key
GXJABQQUPOEUTA-RDJZCZTQSA-N
General description
Application
Biochem/physiol Actions
Features and Benefits
- Cell permeable and enables targeted action
- Allows reversible modulation of cellular processes
Packaging
Warning
Preparation Note
Reconstitution
Other Notes
Tamatani, T., et al. 2013. Int. J. Oncol.42, 935.
Beck, P., et al. 2012. J. Biol. Chem.393, 1101.
Fang, H.T., et al. 2012. Proc. Natl. Acad. Sci. USA.109, 2521.
Chen, D., et al. 2011. Curr. Cancer Drug Targets11, 239.
Demo, S.D., et al. 2007. Cancer Res.67, 6383.
Adams, J., et al. 1999. Cancer Res.59, 2615.
Teicher, B.A., et al. 1999. Clin. Cancer Res.5, 2638.
Adams, J., et al. 1998. Bioorg. Med. Chem. Lett.8, 333.
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Contenido relacionado
Select different protease inhibitor types based on your needs to prevent protein degradation during isolation and characterization and safeguard proteins in sample prep.
Select different protease inhibitor types based on your needs to prevent protein degradation during isolation and characterization and safeguard proteins in sample prep.
Select different protease inhibitor types based on your needs to prevent protein degradation during isolation and characterization and safeguard proteins in sample prep.
Select different protease inhibitor types based on your needs to prevent protein degradation during isolation and characterization and safeguard proteins in sample prep.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico