B22984
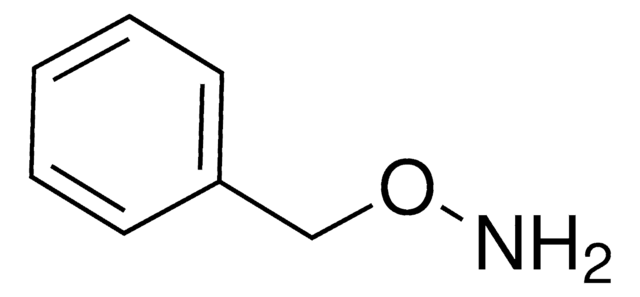
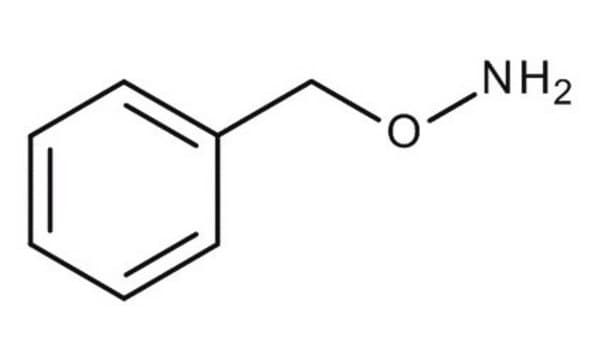
O-Benzylhydroxylamine hydrochloride
99%
Sinónimos:
Benzyloxyamine hydrochloride
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
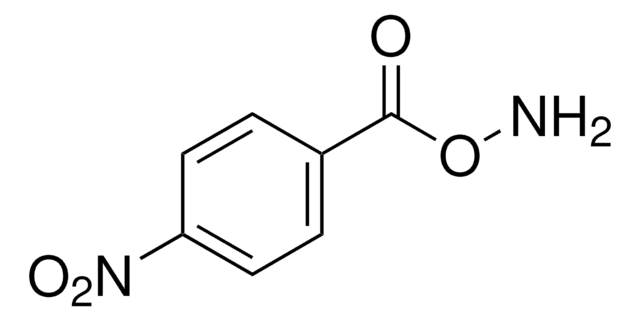
Fórmula lineal:
C6H5CH2ONH2 · HCl
Número de CAS:
Peso molecular:
159.61
Beilstein/REAXYS Number:
3687991
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
99%
form
crystals
mp
238 °C (subl.) (lit.)
SMILES string
Cl.NOCc1ccccc1
InChI
1S/C7H9NO.ClH/c8-9-6-7-4-2-1-3-5-7;/h1-5H,6,8H2;1H
InChI key
HYDZPXNVHXJHBG-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Application
Effective reagent used to prepare α-hydroxybenzylamines from α-hydroxyketones.
signalword
Danger
hcodes
Hazard Classifications
Eye Dam. 1 - Skin Irrit. 2 - Skin Sens. 1
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
G Stampf et al.
Die Pharmazie, 35(1), 43-44 (1980-01-01)
The study of the blood levels and tissue concentrations in mice to which 14C-benzyloxyamine hydrochloride was applied in the form of a spray and of a suspensoid aerosol evidenced the good abosrption of this pharmacon. Maximum blood levels were observed
S M Breckenridge et al.
Journal of chromatography. B, Biomedical sciences and applications, 694(2), 289-296 (1997-07-04)
Extraction and derivatization of carbonyls to benzyloximes, pentafluorobenzyloximes or 2,4-dinitrophenylhydrazones is simplified and reaction times are substantially reduced by simultaneous sorption and derivatization from aqueous solution onto a solid phase. In this reaction a macroreticular polystyrene-divinylbenzene resin acts as a
A A Purmal et al.
Mutation research, 364(3), 193-207 (1996-12-02)
Duplex oligonucleotides containing the base lesion analogs, O-methylhydroxylamine- and O-benzylhydroxylamine-modified abasic (AP) sites, were substrates for the DNA N-glycosylases endonuclease III, formamidopyrimidine DNA N-glycosylase and T4 endonuclease V. These N-glycosylases are known to have associated AP lyase activities. In contrast
[Thermographic analysis of precipitates formed by the interaction of active ingredients and additives].
G Stampf et al.
Acta pharmaceutica Hungarica, 53(6), 268-272 (1983-11-01)
Samia Far et al.
Journal of peptide science : an official publication of the European Peptide Society, 11(7), 424-430 (2005-01-11)
The synthesis of glyoxylyl peptides by coupling the masked glyoxylic acid derivative (FmocNH)(2)CHCO(2)H, 1, to a peptidyl resin assembled using Fmoc/tert-butyl chemistry has been described recently. Deprotection and cleavage of the peptide from the solid support using TFA was followed
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico