93550
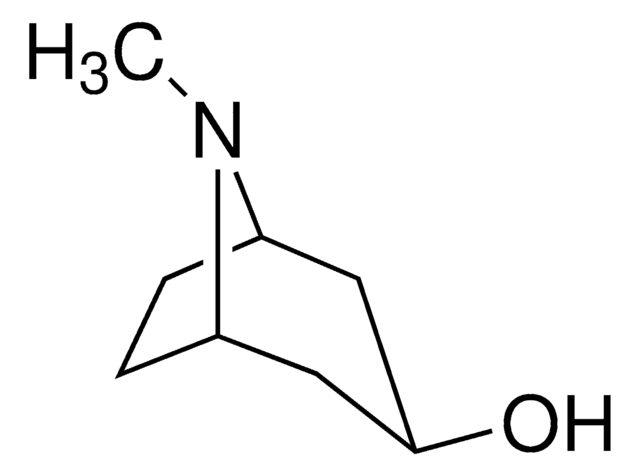
Tropine
≥97.0% (NT)
Sinónimos:
3-Tropanol
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C8H15NO
Número de CAS:
Peso molecular:
141.21
Beilstein/REAXYS Number:
80188
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
≥97.0% (NT)
form
powder
impurities
0-3% water
solubility
H2O: 0.1 g/mL, clear
functional group
hydroxyl
storage temp.
2-8°C
SMILES string
CN1[C@H]2CC[C@@H]1C[C@H](O)C2
InChI
1S/C8H15NO/c1-9-6-2-3-7(9)5-8(10)4-6/h6-8,10H,2-5H2,1H3/t6-,7+,8+
InChI key
CYHOMWAPJJPNMW-JIGDXULJSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Birgit Dräger
Phytochemistry, 67(4), 327-337 (2006-01-24)
Two stereospecific oxidoreductases constitute a branch point in tropane alkaloid metabolism. Products of tropane metabolism are the alkaloids hyoscyamine, scopolamine, cocaine, and polyhydroxylated nortropane alkaloids, the calystegines. Both tropinone reductases reduce the precursor tropinone to yield either tropine or pseudotropine.
A Yamashita et al.
Biochemistry, 38(24), 7630-7637 (1999-07-01)
Tropinone reductase-II (TR-II) catalyzes the NADPH-dependent reduction of the carbonyl group of tropinone to a beta-hydroxyl group. The crystal structure of TR-II complexed with NADP+ and pseudotropine (psi-tropine) has been determined at 1.9 A resolution. A seven-residue peptide near the
Gábor Maksay et al.
Bioorganic & medicinal chemistry, 17(19), 6872-6878 (2009-09-04)
Heteroaromatic carboxylic esters of (nor)tropine were synthesized. Tropine esters displaced [(3)H]strychnine binding to glycine receptors of rat spinal cord with low Hill slopes. Two-site displacement resulted in nanomolar IC(50,1) and micromolar IC(50,2) values, and IC(50,2)/IC(50,1) ratios up to 615 depending
Laszlo Gyermek et al.
Journal of critical care, 24(1), 58-65 (2009-03-11)
There is a need for neuromuscular relaxant (NMR) agents that are of the "nondepolarizing type" and produce rapidly developing and short-lasting skeletal muscle relaxation in anesthesiology. Many efforts have been directed to produce such agents. Our research focused on the
Renata A Kwiecień et al.
Archives of biochemistry and biophysics, 510(1), 35-41 (2011-03-23)
(15)N heavy isotope effects are especially useful when detail is sought pertaining to the reaction mechanism for the cleavage of a C-N bond. Their potential in assisting to describe the mechanism of N-demethylation of tertiary amines by the action of
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico