139351
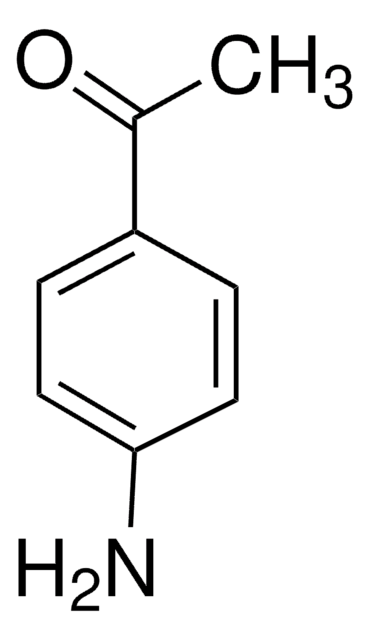
3′-Aminoacetophenone
97%
Sinónimos:
3-Acetylaniline
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
H2NC6H4COCH3
Número de CAS:
Peso molecular:
135.16
Beilstein/REAXYS Number:
386009
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
Quality Level
assay
97%
bp
289-290 °C (lit.)
mp
94-98 °C (lit.)
functional group
ketone
SMILES string
CC(=O)c1cccc(N)c1
InChI
1S/C8H9NO/c1-6(10)7-3-2-4-8(9)5-7/h2-5H,9H2,1H3
InChI key
CKQHAYFOPRIUOM-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
General description
3′-Aminoacetophenone acts as bifunctional coupling reagent during the synthesis of pyrimidines.
Application
3′-Aminoacetophenone (3-Acetylaniline) was used as reagent during the asymmetric total synthesis of pactamycin. It was used as starting reagent during the synthesis of curcumin mimics with substituted sulfonyl group.
signalword
Warning
hcodes
Hazard Classifications
Acute Tox. 4 Oral
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Chan Mug Ahn et al.
Bioorganic & medicinal chemistry letters, 19(5), 1481-1483 (2009-01-31)
In order to discover novel small vasodilatory molecules for potential use in the treatment of vascular disease, we tested the vasodilatation effect of two types of synthetic curcumin mimics, amide type (3) and sulfonyl amide type (4), upon the basilar
J S Früchtel et al.
Biotechnology and bioengineering, 71(2), 94-103 (2001-04-05)
A combination of symmetric building blocks and combinatorial functional group transformation for synthesis of pyrimidines was investigated. The purpose of the study was to maximize the return on invested synthetic efforts of reaction development for libraries. A representative set of
M K Subramanian et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 71(1), 59-67 (2008-01-08)
The gas phase infrared spectrum of 3-aminoacetophenone (3AAP) was measured in the range 5000-500 cm(-1) and with a resolution of 0.5 cm(-1). The Fourier transform Raman (FT-Raman) and Fourier transform infrared (FT-IR) spectra of 3AAP were recorded in the solid
Justin T Malinowski et al.
Science (New York, N.Y.), 340(6129), 180-182 (2013-04-13)
Medicinal application of many complex natural products is precluded by the impracticality of their chemical synthesis. Pactamycin, the most structurally intricate aminocyclopentitol antibiotic, displays potent antiproliferative properties across multiple phylogenetic domains, but it is highly cytotoxic. A limited number of
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico