All Photos(4)
About This Item
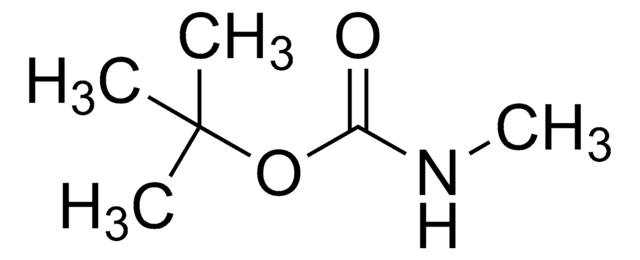
Linear Formula:
CH3C6H4SO2NHCO2C(CH3)3
CAS Number:
Molecular Weight:
271.33
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
mp
121-123 °C (lit.)
solubility
chloroform: soluble 25 mg/mL, clear, colorless
SMILES string
Cc1ccc(cc1)S(=O)(=O)NC(=O)OC(C)(C)C
InChI
1S/C12H17NO4S/c1-9-5-7-10(8-6-9)18(15,16)13-11(14)17-12(2,3)4/h5-8H,1-4H3,(H,13,14)
InChI key
DUTLOVSBVBGNDM-UHFFFAOYSA-N
General description
N-(tert-Butoxycarbonyl)-p-toluenesulfonamide is a N-substituted sulphonamide and its reaction with N-trityl L-serine esters under Mitsunobu reaction conditions is reported. It can be directly coupled with primary, secondary and allylic alcohols under Mitsunobu conditions to afford various sulfonyl-protected amines.
Application
N-(tert-Butoxycarbonyl)-p-toluenesulfonamide may be used in the preparation of enyne amide, precursor for Pauson-Khand reaction.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Toshio Honda et al.
The Journal of organic chemistry, 72(17), 6541-6547 (2007-07-31)
Diastereoselective formal synthesis of a monoterpene alkaloid, (-)-incarvilline, the key intermediate for the synthesis of (-)-incarvillateine, was achieved by using an intramolecular Pauson-Khand reaction of (S)-N-[(E)-2-butenyl]-N-(3-butynyl-2-methoxymethoxy)-p-toluenesulfonamide as a key step.
Tetrahedron Letters, 30, 5709-5709 (1989)
Use of the Mitsunobu reaction in the synthesis of orthogonally protected a, ?-diaminopropionic acids.
Kelleher F.
Tetrahedron Letters, 48(28), 4879-4882 (2007)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service