560529
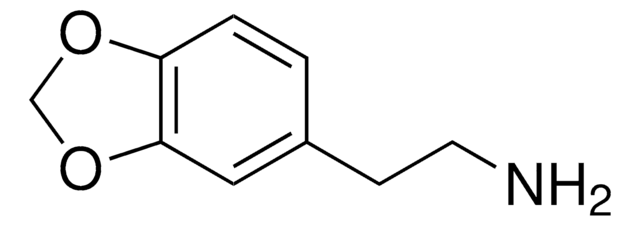
3,4-Methylenedioxyphenethylamine hydrochloride
98%
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C9H11NO2 · HCl
CAS Number:
Molecular Weight:
201.65
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
mp
216-218 °C (lit.)
functional group
amine
SMILES string
Cl.NCCc1ccc2OCOc2c1
InChI
1S/C9H11NO2.ClH/c10-4-3-7-1-2-8-9(5-7)12-6-11-8;/h1-2,5H,3-4,6,10H2;1H
InChI key
NDYXFQODWGEGNU-UHFFFAOYSA-N
General description
3,4-Methylenedioxyphenethylamine hydrochloride can be synthesized by reacting aluminum chloride, LiAlH4 and 3,4-methylenedioxyphenylacetonitrile.
Application
3,4-Methylenedioxyphenethylamine hydrochloride may be used to synthesize N-(3,4-methylenedioxyphenethyl)-2-(3-bromo-4-methoxyphenyl)acetamide.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
J F Bagli et al.
Journal of medicinal chemistry, 19(7), 876-882 (1976-07-01)
Synthesis of a series of thienylethanolamines having varying substituents on the thiophene ring and on the nitrogen atom is described using the general procedure reported earlier. In the determination of their pharmacological profile, some of the derivatives showed marked antihypertensive
Milica Ninković et al.
Nephrology (Carlton, Vic.), 13(1), 33-37 (2008-01-18)
The mechanism of MDMA (3,4-methylenedioxymethamphetamine)-induced toxicity is believed to be, in part, due to enhanced oxidative stress. As MDMA is eliminated via the kidney, the aim of this study was to investigate whether MDMA created conditions of oxidative stress within
Laura Aalberg et al.
Journal of chromatographic science, 41(5), 227-233 (2003-07-05)
Three regioisomeric 3,4-methylenedioxyphenethylamines having the same molecular weight and major mass spectral fragments of equivalent mass have been reported as components of clandestine drug samples in recent years. These drugs of abuse are 3,4-methylenedioxy-N-ethylamphetamine, 3,4-methylenedioxy-N,N-dimethylamphetamine, and N-methyl-1-(3,4-methylenedioxyphenyl)-2-butanamine. These three compounds
D J De Silva et al.
Neuroscience, 134(4), 1363-1375 (2005-08-02)
Substituted amphetamines such as p-chloroamphetamine and the abused drug methylenedioxymethamphetamine cause selective destruction of serotonin axons in rats, by unknown mechanisms. Since some serotonin neurones also express neuronal nitric oxide synthase, which has been implicated in neurotoxicity, the present study
An aryne route to laureline, and related topics.
Gibson MS, et al.
J. Chem. Soc. Sect. C, 16, 2234-2238 (1970)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service