SML2706
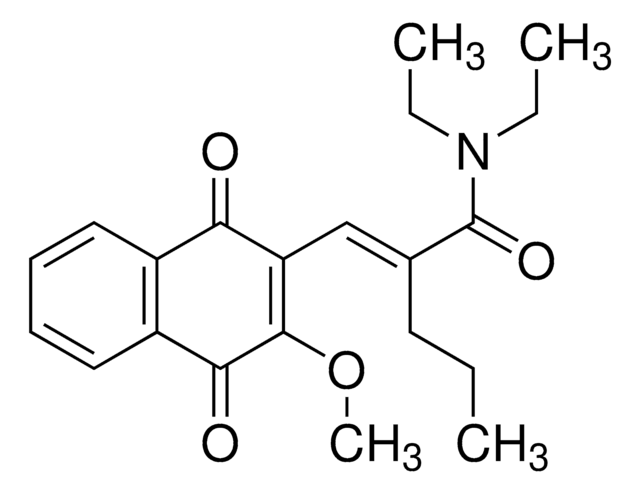
Jadomycin B
≥98% (HPLC)
Sinónimos:
1-(sec-butyl)-12-((4,5-dihydroxy-6-methyltetrahydro-2H-pyran-2-yl)oxy)-7-hydroxy-5-methyl-8H-benzo[b]oxazolo[3,2-f]phenanthridine-2,8,13(1H,3aH)-trione
About This Item
Productos recomendados
biological source
Streptomyces venezuelae
Quality Level
assay
≥98% (HPLC)
form
powder
storage condition
protect from light
solubility
DMSO: 1 mg/mL
storage temp.
−20°C
InChI
1S/C30H31NO9/c1-5-13(3)24-30(37)40-29-16-9-12(2)10-17(32)21(16)23-25(31(24)29)28(36)22-15(27(23)35)7-6-8-19(22)39-20-11-18(33)26(34)14(4)38-20/h6-10,13-14,18,20,24,26,29,32-34H,5,11H2,1-4H3
InChI key
BSBSCJRAEMDCHC-UHFFFAOYSA-N
General description
Jadomycin B displays antimicrobial, anti-tumor, aurora-B kinase inhibition, DNA cleaving and more activities.3,4,5,6
Jadomycin B was found to be active against a variety of staphylococci, including methicillin-resistant Staphylococcus aureus in a MIC of 1μg/ml.3 In addition, its anti-tumor activity was demonstrated as it kills drug-sensitive and multidrug-resistant breast cancer cell, through inhibition of type II topoisomerases and the induction of DNA damage and apoptosis. Jadomycin B (15 mM), 24-hour treatment significantly lowered the levels of topoisomerase IIa protein versus the vehicle control.4
It was also shown that Jadomycin B inhibits Aurora-B kinase activity by phosphorylation of histone H3 on Ser10 in a dose-dependent manner (10μg /mL Jadomycin B reduced H3 phosphorylation by 70%).5
Jadomycin B was also found to cleave DNA in the presence of Cu (II) by reducing it to Cu(I) which can further react with H2O2 to form hydroxyl radicals that causes DNA strand scission without the requirement of any external reducing agent. The EC50 value of Jadomycin B for single-strand scission was approximately 1.7μM.6
Biochem/physiol Actions
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico





![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)