Iniciar sesión para ver los precios por organización y contrato.
Acerca de este artículo
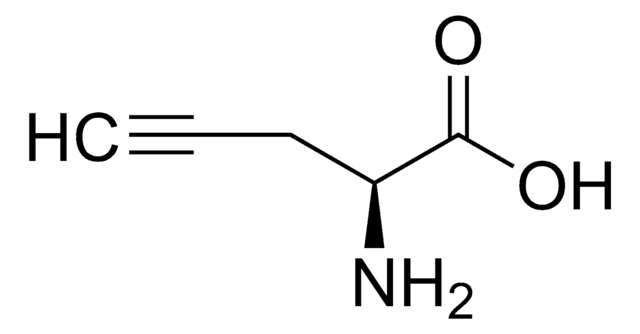
Fórmula empírica (notación de Hill):
C5H7NO2
Número CAS:
Peso molecular:
113.11
UNSPSC Code:
12352209
eCl@ss:
32160406
PubChem Substance ID:
NACRES:
NA.26
Beilstein/REAXYS Number:
2347861
MDL number:
Nombre del producto
L-C-Propargylglycine, ≥99.0% (TLC)
assay
≥99.0% (TLC)
form
powder
color
white
mp
235-239 °C
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
N[C@@H](CC#C)C(O)=O
InChI
1S/C5H7NO2/c1-2-3-4(6)5(7)8/h1,4H,3,6H2,(H,7,8)/t4-/m0/s1
InChI key
DGYHPLMPMRKMPD-BYPYZUCNSA-N
Application
Biochem/physiol Actions
L-C-Propargylglycine, a specific inhibitor of H(2)S synthase of cystathionine-γ-lyase (CSE), may be used to study the role of H2S in regulation of biological processes.
L-propargylglycine (PAG), an inhibitor of cystathionine γ-lyase (CSE), is useful in studies of hydrogen sulphide synthesis and bioactivity.
1 of 1
Este artículo | |||
|---|---|---|---|
| form powder | form powder | form crystals | form powder |
| assay ≥99.0% (TLC) | assay ≥98% (TLC) | assay ≥98.0% (HPLC) | assay ≥98.0% (TLC) |
| storage temp. 2-8°C | storage temp. −20°C | storage temp. −20°C | storage temp. 2-8°C |
| mp 235-239 °C | mp - | mp - | mp 76-80 °C |
| color white | color white | color - | color - |
| application(s) peptide synthesis | application(s) cell analysis | application(s) peptide synthesis | application(s) peptide synthesis |
Clase de almacenamiento
11 - Combustible Solids
wgk
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.