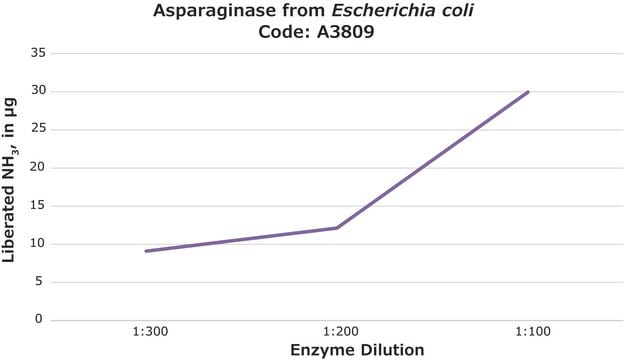
The activity and protein concentration of this product varies from lot to lot and are reported in the product Certificate of Analysis. This information may then be used to determine the lot specific volume. As an example, a lot reporting a 99 mg/mL protein concentration would result in a volume of 2.53 mL for the 250 mg package size. This 250 mg package would have a total activity of 2436 units. Please see the link below to review a sample or lot specific Certificate:
https://www.sigmaaldrich.com/product/sigma/76427#product-documentation
Seleccione un Tamaño
| A ustedes/SKU | Disponibilidad | Precio |
|---|---|---|
250 mg | Envío estimado HOYdesdeSAINT LOUIS | $198.00 |
1 g | Envío estimado HOYdesdeSAINT LOUIS | $566.00 |
Acerca de este artículo
$198.00
Envío estimado HOYDetalles
biological source
Escherichia coli
Quality Segment
form
suspension
specific activity
5-10 units/mg protein
mol wt
Mr ~70000
technique(s)
activity assay: suitable
application(s)
diagnostic assay manufacturing
storage temp.
2-8°C
General description
Application
Biochem/physiol Actions
Other Notes
1 of 1
Este artículo | |||
|---|---|---|---|
| specific activity 5-10 units/mg protein | specific activity ≥50 units/mg protein | specific activity 30-90 units/mg protein (modified Warburg-Christian, in glycine buffer) | specific activity 100-300 units/mg protein (biuret) |
| technique(s) activity assay: suitable | technique(s) - | technique(s) - | technique(s) - |
| biological source Escherichia coli | biological source Escherichia coli | biological source Escherichia coli | biological source - |
| application(s) diagnostic assay manufacturing | application(s) - | application(s) - | application(s) - |
| form suspension | form lyophilized powder | form ammonium sulfate suspension | form lyophilized powder |
| mol wt Mr ~70000 | mol wt 110 kDa | mol wt - | mol wt - |
Clase de almacenamiento
11 - Combustible Solids
wgk
WGK 1
flash_point_f
Not applicable
flash_point_c
Not applicable
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
-
Good afternoon, I recently received Penicillin-Amidase aus E. coli 5-10 units/mg protein (250mg). Could you please guide me how much volume does it contain so that we can calculate the precise amount of concenteration available. Thank you in advance.
1 answer-
Helpful?
-