If this product has an expiration or retest date, it will be shown on the Certificate of Analysis (COA, CofA). If there is no retest or expiration date listed on the product's COA, we do not have suitable stability data to determine a shelf life. For these products, the only date on the COA will be the release date; a retest, expiration, or use-by-date will not be displayed.
For all products, we recommend handling per defined conditions as printed in our product literature and website product descriptions. We recommend that products should be routinely inspected by customers to ensure they perform as expected.
For products without retest or expiration dates, our standard warranty of 1 year from the date of shipment is applicable.
For more information, please refer to the Product Dating Information document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/449/386/product-dating-information-mk.pdf
38370
DIC
≥98.0% (GC), for peptide synthesis
Sinónimos:
N,N′-Diisopropilcarbodiimida
Seleccione un Tamaño
Seleccione un Tamaño
About This Item
Productos recomendados
Nombre del producto
DIC, purum, ≥98.0% (GC)
grado
purum
Nivel de calidad
Ensayo
≥98.0% (GC)
Formulario
liquid
idoneidad de la reacción
reaction type: Coupling Reactions
índice de refracción
n20/D 1.433 (lit.)
bp
145-148 °C (lit.)
densidad
0.815 g/mL at 20 °C (lit.)
0.815 g/mL at 20 °C
aplicaciones
peptide synthesis
grupo funcional
amine
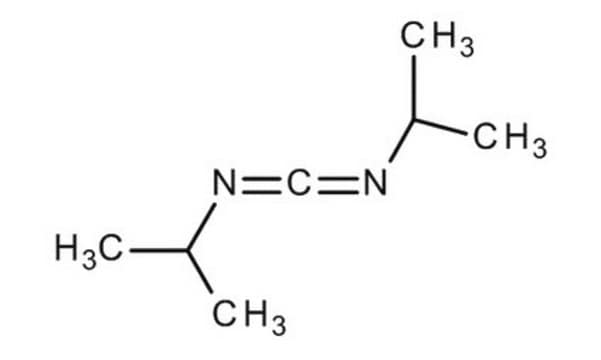
cadena SMILES
CC(C)N=C=NC(C)C
InChI
1S/C7H14N2/c1-6(2)8-5-9-7(3)4/h6-7H,1-4H3
Clave InChI
BDNKZNFMNDZQMI-UHFFFAOYSA-N
Información sobre el gen
human ... EPHX2(2053)
mouse ... Ephx2(13850)
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
Aplicación
- To synthesize lanthanide (Ln) guanidinate complexes via insertion of carbodiimide into the Ln-N bond of lanthanocene secondary amido complexes.[2]
- To facilitate the cyclization of N-(β-Hydroxy)amides to form 2-oxazolines.[3]
- To synthesize 1-isopropyl-2-alkoxycarbonyl-3-isopropyliminio-aziridine by reacting with alkyl diazoacetates in the presence of transition metal salts.[4]
- A coupling reagent for the synthesis of various esters and amides by treating carboxylic acids with phenols and amines respectively.[5]
- A reagent for the conversion of alcohols to aldehydes or ketones in the presence of DMSO via modified Moffatt-type oxidation reaction.[1]
- A reagent to facilitates the preparation of alkyl halides from corresponding alcohols via the formation of o-alkylisourea.[6]
Producto relacionado
Palabra de señalización
Danger
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Acute Tox. 1 Inhalation - Eye Dam. 1 - Flam. Liq. 3 - Resp. Sens. 1 - Skin Sens. 1
Código de clase de almacenamiento
3 - Flammable liquids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
91.4 °F
Punto de inflamabilidad (°C)
33 °C
Equipo de protección personal
Eyeshields, Faceshields, Gloves, type ABEK (EN14387) respirator filter
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
-
How can I determine the shelf life / expiration / retest date of this product?
1 answer-
Helpful?
-
-
How is shipping temperature determined? And how is it related to the product storage temperature?
1 answer-
Products may be shipped at a different temperature than the recommended long-term storage temperature. If the product quality is sensitive to short-term exposure to conditions other than the recommended long-term storage, it will be shipped on wet or dry-ice. If the product quality is NOT affected by short-term exposure to conditions other than the recommended long-term storage, it will be shipped at ambient temperature. As shipping routes are configured for minimum transit times, shipping at ambient temperature helps control shipping costs for our customers. For more information, please refer to the Storage and Transport Conditions document: https://www.sigmaaldrich.com/deepweb/assets/sigmaaldrich/marketing/global/documents/316/622/storage-transport-conditions-mk.pdf
Helpful?
-
-
hi, what is the concentration of this product?
1 answer-
The concentration of this product can be determined using the lot specific purity reported in the Certificate of Analysis, the density, and the compound molecular weight. This product has a density of 0.815 g/mL and a molecular weight of 126.20 g/mol. The minimum purity is 98.0%, thus the molarity will vary slightly from lot to lot. The molarity can range from 6.33M (98.0%) to 6.46M (100%).
Helpful?
-
Active Filters
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico