78194
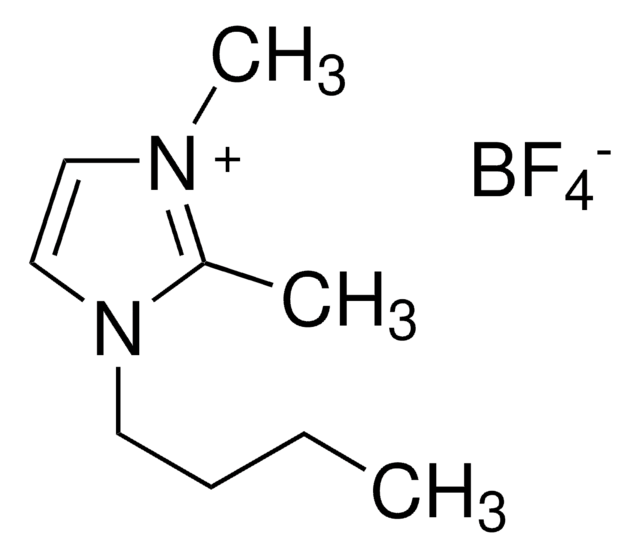
1-Butyl-2,3-dimethylimidazolium chloride
≥97.0% (HPLC/AT)
About This Item
Productos recomendados
assay
≥97.0% (HPLC/AT)
form
crystals
impurities
≤1.0% water
SMILES string
[Cl-].CCCCn1cc[n+](C)c1C
InChI
1S/C9H17N2.ClH/c1-4-5-6-11-8-7-10(3)9(11)2;/h7-8H,4-6H2,1-3H3;1H/q+1;/p-1
InChI key
HHHYPTORQNESCU-UHFFFAOYSA-M
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Application
- As a solvent in the chemical modification of polysaccharide cellulose.
- As a model ionic liquid in the conversion of a monosaccharide like fructose into 5-hydroxymethylfurfural using H2SO4.
- To prepare 1-butyl-2,3-dimethylimidazolium dicarba-7,8-nidoundecaborate by reacting with caesium dicarba-7,8-nido-undecaborate.
- To prepare mesoporous ZnAl2O4 nanomaterials, which are used as catalysts or catalyst supports.
Physical form
Other Notes
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Functionalized imidazolium cations with thioether, urea, or thiourea derivatized side chains act as metal-ligating moieties, whereas the PF6– anions provide the desired water immiscibility.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico