513997
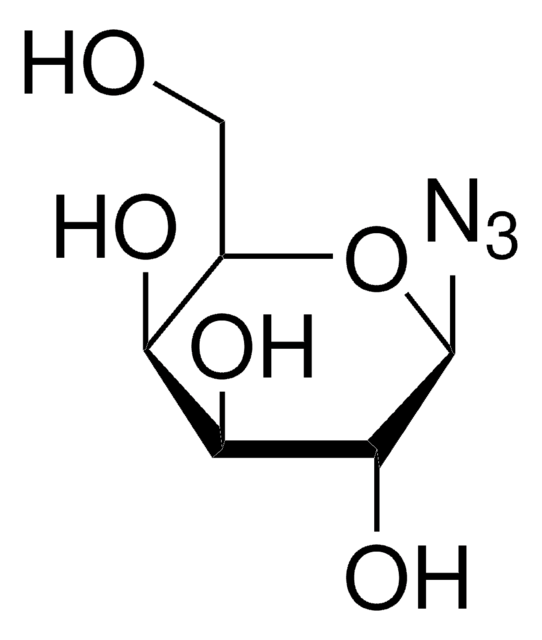
1-Azido-1-deoxy-β-D-glucopyranoside tetraacetate
Sinónimos:
NSC 272456
About This Item
Productos recomendados
form
solid
optical activity
[α]/D -29°, c = 1% in H2O
[α]/D -30°, c = 1% in chloroform
reaction suitability
reaction type: click chemistry
mp
127-131 °C (lit.)
SMILES string
CC(=O)OC[C@H]1O[C@@H](N=[N+]=[N-])[C@H](OC(C)=O)[C@@H](OC(C)=O)[C@H]1OC(C)=O
InChI
1S/C14H19N3O9/c1-6(18)22-5-10-11(23-7(2)19)12(24-8(3)20)13(25-9(4)21)14(26-10)16-17-15/h10-14H,5H2,1-4H3/t10-,11+,12+,13-,14-/m1/s1
InChI key
NHNYHKRWHCWHAJ-MBJXGIAVSA-N
Application
- 1,2,3-Triazole-boron dipyrromethenes (BODIPYs) containing glucose groups via Cu(I)-catalyzed azide–alkyne ″click″ cycloaddition reaction conditions.
- 1-(β-D-glycosyl)-5-benzenesulfonamide-1,2,3-triazole derivatives by ruthenium-catalyzed azide-alkyne cycloaddition reactions.
- 2,3,4,6-Tetra-O-acetyl-β-D-glucopyranosylamine by palladium catalyzed hydrogenation reaction.
- Glycoside annulated dihydropyrimidinone derivatives by one-pot five-component condensation reaction with tert-butyl β-ketoester, arylaldehyde, urea and propargyl alcohol.
- Synthesis of Protein Tyrosine Phosphatase 1B inhibitor
- Synthesis of glycoconjugate carbonic anhydrase inhibitors by ruthenium-catalyzed azide-alkyne 1,3-dipolar cycloaddition
- Synthesis of variously coupled conjugates of D-glucose via click chemistry for inhibition of glycogen phosphorylase
- Hydrogenation reactions
- Preparation of posttranslationally modified peptides efficiently mimicking neoantigens in relation to autoimmune disease
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
flash_point_f
Not applicable
flash_point_c
Not applicable
ppe
Eyeshields, Gloves, type N95 (US)
Elija entre una de las versiones más recientes:
Certificados de análisis (COA)
¿No ve la versión correcta?
Si necesita una versión concreta, puede buscar un certificado específico por el número de lote.
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico