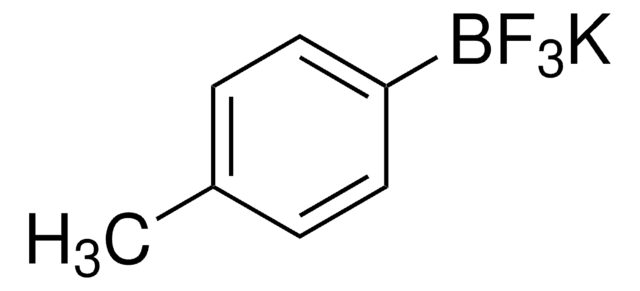
704415
Vinylboronic acid MIDA ester
97%
Synonym(s):
6-Methyl-2-vinyl-1,3,6,2-dioxazaborocane-4,8-dione
Sign Into View Organizational & Contract Pricing
All Photos(4)
About This Item
Empirical Formula (Hill Notation):
C7H10BNO4
CAS Number:
Molecular Weight:
182.97
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
powder
mp
152-156 °C
storage temp.
2-8°C
SMILES string
CN1CC(=O)OB(OC(=O)C1)C=C
InChI
1S/C7H10BNO4/c1-3-8-12-6(10)4-9(2)5-7(11)13-8/h3H,1,4-5H2,2H3
InChI key
MGRQGYAVASCCAK-UHFFFAOYSA-N
Related Categories
General description
Vinylboronic acid MIDA ester, like other MIDA boronates, possesses the capacity for controlled, in situ slow-release of boronic acids under aqueous basic conditions allowing the cross-coupling of classically challenging substrates.
Application
MIDA boronates as stable boronic acid surrogates for classically challenging cross-couplings
Suzuki Cross-Coupling with MIDA Boronates
Suzuki Cross-Coupling with MIDA Boronates
- Vinylboronic acid MIDA ester is an air and chromatographically stable boronic acid surrogate for Suzuki-Miyaura cross-coupling. It can also be used in Heck and oxidative Heck reactions as well as in olefin metathesis to provide the cross-coupled product.
- It is compatible with a wide range of common synthetic reagents that allows functionalization to synthesize structurally complex boronic acid surrogates.
- It undergoes cyclopropanation and epoxidation to yield corresponding MIDA cyclopropane and oxirane, respectively.
- It can be used as one of the major reagents for the scalable synthesis of potent cytotoxin, Leiodermatolide and for the total synthesis of (−)-Blepharocalyxin D.
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis, molecular editing, and biological assessment of the potent cytotoxin leiodermatolide.
Mailhol D, et al.
Journal of the American Chemical Society, 136(44), 15719-15729 (2014)
(1-Bromovinyl)-MIDA boronate: a readily accessible and highly versatile building block for small molecule synthesis.
Woerly EM, et al.
Tetrahedron, 69(36), 7732-7740 (2013)
Vinyl MIDA boronate: a readily accessible and highly versatile building block for small molecule synthesis.
Uno BE, et al.
Tetrahedron, 65(16), 3130-3138 (2009)
Synthesis of trans-2-(Trifluoromethyl) cyclopropanes via Suzuki reactions with an N-methyliminodiacetic acid boronate.
Duncton MA and Singh R.
Organic Letters, 15(17), 4284-4287 (2013)
A general solution for unstable boronic acids: slow-release cross-coupling from air-stable MIDA boronates.
Knapp DM, et al.
Journal of the American Chemical Society, 131(20), 6961-6963 (2009)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service




![[1,1′-Bis(diphenylphosphino)ferrocene]dichloropalladium(II)](/deepweb/assets/sigmaaldrich/product/structures/130/734/8846aa26-1858-458a-998d-8c306c13bf0f/640/8846aa26-1858-458a-998d-8c306c13bf0f.png)