903701
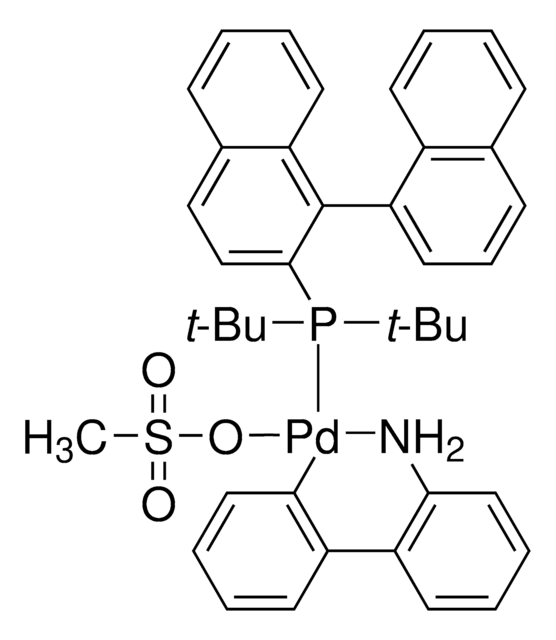
tBuDavePhos Pd G3
1:1 THF adduct
Synonym(s):
t-BuDavePhos G3 Palladacycle, t-BuDavePhos Palladacycle
About This Item
Recommended Products
form
powder or crystals
feature
generation 3
reaction suitability
core: palladium
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reaction type: Heck Reaction
reaction type: Hiyama Coupling
reaction type: Negishi Coupling
reaction type: Sonogashira Coupling
reaction type: Stille Coupling
reaction type: Suzuki-Miyaura Coupling
reagent type: catalyst
reaction type: Cross Couplings
functional group
phosphine
SMILES string
CN(C1=C(C2=C(P(C(C)(C)C)C(C)(C)C)C=CC=C2)C=CC=C1)C.NC3=C(C4=C([Pd]OS(C)(=O)=O)C=CC=C4)C=CC=C3.C5COCC5
General description
Application
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service