T69000
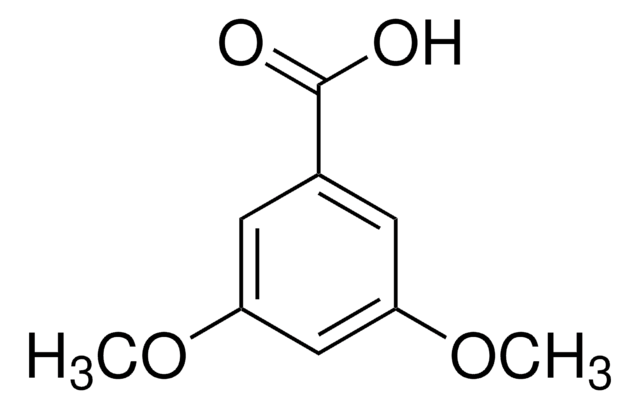
3,4,5-Trimethoxybenzoic acid
ReagentPlus®, 99%
Synonym(s):
Gallic acid trimethyl ether, Trimethylgallic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
(CH3O)3C6H2CO2H
CAS Number:
Molecular Weight:
212.20
Beilstein:
884655
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
product line
ReagentPlus®
Assay
99%
bp
225-227 °C/10 mmHg (lit.)
mp
168-171 °C (lit.)
SMILES string
COc1cc(cc(OC)c1OC)C(O)=O
InChI
1S/C10H12O5/c1-13-7-4-6(10(11)12)5-8(14-2)9(7)15-3/h4-5H,1-3H3,(H,11,12)
InChI key
SJSOFNCYXJUNBT-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Legal Information
ReagentPlus is a registered trademark of Merck KGaA, Darmstadt, Germany
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
E N Schachter et al.
Respiration; international review of thoracic diseases, 65(5), 393-400 (1998-10-23)
Latex manufacturing workers are exposed to a heterogeneous aerosol of organic compounds. Previous studies of latex workers involved in glove production indicate that these individuals are at risk of developing respiratory symptoms and impaired lung function. The effect of latex
H Naviasky
Journal of pharmaceutical sciences, 73(4), 542-545 (1984-04-01)
An ion-pair column chromatographic/UV spectrophotometric method for assaying trimethobenzamide hydrochloride in capsules and injections is presented, as well as a method for the detection of 3,4,5- trimethoxybenzoic acid in trimethobenzamide hydrochloride bulk drug and dosage forms. Results obtained by the
M I Donnelly et al.
Journal of bacteriology, 142(3), 916-924 (1980-06-01)
When grown at the expense of 3,4,5-trimethoxybenzoic acid, a strain of Pseudomonas putida oxidized this compound and also 3,5-dimethoxy-4-hydroxybenzoic (syringic) and 3,4-dihydroxy-5-methoxybenzoic (3-O-methylgallic) acids; but other hydroxy- or methoxy-benzoic acids were oxidized slowly or not at all. Radioactivity appeared exclusively
Y Mimaki et al.
Bioscience, biotechnology, and biochemistry, 60(6), 1049-1050 (1996-06-01)
Bioassay-guided fractionation of the MeOH extract of Ornithogalum saundersiae bulbs led to the isolation of a new cholestane bisdesmoside with potent cytotoxic activities toward leukemia HL-60 and MOLT-4 cells. The structure was deduced mainly from spectroscopic information.
H Tateno et al.
Mutation research, 327(1-2), 237-246 (1995-03-01)
Many inhibitors of tubulin polymerization have a trimethoxybenzene ring in their molecules. Such trimethoxybenzoic compounds and their analogues may therefore have a potency to induce meiotic nondisjunction of oocytes. In this study, a single dose of reserpine (0.5 microgram/g body
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service