A77997
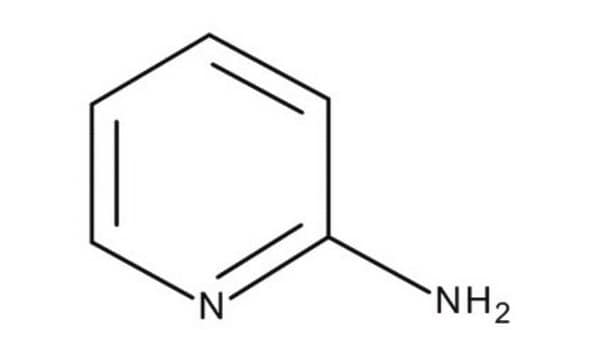
2-Aminopyridine
99%
Synonym(s):
2-AP, 2-Pyridinamine, 2-Pyridylamine
About This Item
Recommended Products
Quality Level
Assay
99%
form
crystals
bp
204-210 °C (lit.)
mp
54-58 °C (lit.)
SMILES string
Nc1ccccn1
InChI
1S/C5H6N2/c6-5-3-1-2-4-7-5/h1-4H,(H2,6,7)
InChI key
ICSNLGPSRYBMBD-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
It can also be used:
- As a reactant in the synthesis of 3-aroylimidazopyridines from chalcones by aerobic oxidative amidation using copper acetate catalyst.
- In the synthesis of crystalline Cu(II) complex, di-μ-(2-aminopyridine(N,N′))-bis[(2,6 pyridinedicarboxylate)aquacopper(II)] tetrahydrate using 2,6-pyridinedicarboxylic acid and Cu(CH3COO)2.H2O.
- As an imprinting molecule for the preparation of poly(methacrylic acid–ethylene glycol dimethacrylate) polymer. It is packed in micro-column for selective solid phase extraction of 2-aminopyridine.
- As a reactant in the synthesis of 2-aryl-3-(pyridin-2-yl)-1,3-thiazolidin-4-ones in the presence of Lewis acid catalysts.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 3 Dermal - Acute Tox. 3 Oral - Aquatic Chronic 3 - Eye Dam. 1 - Skin Corr. 1A
Storage Class Code
6.1A - Combustible acute toxic Cat. 1 and 2 / very toxic hazardous materials
WGK
WGK 3
Flash Point(F)
197.6 °F - closed cup
Flash Point(C)
92 °C - closed cup
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Explore mass spectrometry analysis of glycans for glycomic & glycoproteomic neutral & acidic glycan analysis. See a general mass spec glycan analysis procedure.
Explore mass spectrometry analysis of glycans for glycomic & glycoproteomic neutral & acidic glycan analysis. See a general mass spec glycan analysis procedure.
Explore mass spectrometry analysis of glycans for glycomic & glycoproteomic neutral & acidic glycan analysis. See a general mass spec glycan analysis procedure.
Explore mass spectrometry analysis of glycans for glycomic & glycoproteomic neutral & acidic glycan analysis. See a general mass spec glycan analysis procedure.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service