T30201
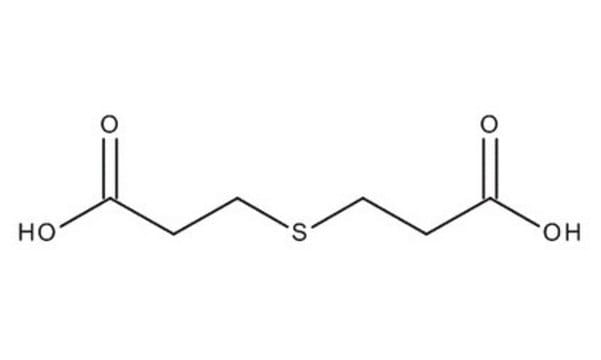
3,3′-Thiodipropionic acid
97%
Synonym(s):
Bis(2-carboxyethyl) sulfide
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Linear Formula:
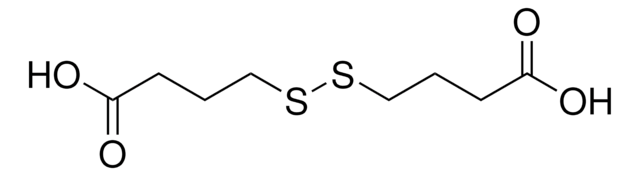
S(CH2CH2COOH)2
CAS Number:
Molecular Weight:
178.21
Beilstein:
1210299
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
mp
131-134 °C (lit.)
SMILES string
OC(=O)CCSCCC(O)=O
InChI
1S/C6H10O4S/c7-5(8)1-3-11-4-2-6(9)10/h1-4H2,(H,7,8)(H,9,10)
InChI key
ODJQKYXPKWQWNK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
3,3′-Thiodipropionic acid is an organic disulfide that can be used as a ligand for the synthesis of cadminum and zinc coordination polymers with luminescent properties.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2
Storage Class Code
11 - Combustible Solids
WGK
WGK 1
Flash Point(F)
262.4 °F - closed cup
Flash Point(C)
128 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Nadine Bruland et al.
The Journal of biological chemistry, 284(1), 660-672 (2008-11-13)
The thioether 3,3-thiodipropionic acid can be used as precursor substrate for biotechnological synthesis of 3-mercaptopropionic acid-containing polythioesters. Therefore, the hitherto unknown catabolism of this compound was elucidated to engineer novel and improved polythioester biosynthesis pathways in the future. Bacteria capable
Yasutaka Kamei et al.
Macromolecular bioscience, 7(3), 364-372 (2007-03-21)
To prepare sulfur-containing natural polymers effectively, several plant oils and 3,3'-thiodipropionic acid (TDP) have been used as carbon sources for the biosynthesis of copolymer poly[(3-hydroxybutyrate)-co-(3-mercaptopropionate)] [poly(3HB-co-3MP)] by a wild-type bacterium Cupriviadus necator H16. By using the plant oils, copolymer accumulation
L C McDonald et al.
Applied and environmental microbiology, 45(2), 360-365 (1983-02-01)
Escherichia coli LSUFS was injured either by freezing at -10 degrees C or by heating at 57 degrees C for 12 min. Surviving cells were recovered on nonselective tryptone-glucose extract agar and selective violet red bile agar supplemented with compounds
E Fehling et al.
Biotechnology and bioengineering, 99(5), 1074-1084 (2007-10-31)
Linear copolymeric polyesters (polyoxoesters) containing thioether functions [poly(3,3'-thiodipropionic acid-co-alpha,omega-alkanediols)] were formed in good yield by esterification of an equimolar mixture of 3,3'-thiodipropionic acid (4-thiaheptane-1,7-dioic acid) and 1,6-hexanediol (weight average molecular mass, M(W) >600 Da: approximately 81% after 6 h) or
Spontaneously organized molecular assemblies. 3. Preparation and properties of solution adsorbed monolayers of organic disulfides on gold surfaces.
Nuzzo R, et al.
Journal of the American Chemical Society, 109(8), 2358-2368 (1987)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service