All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C12H9NO
CAS Number:
Molecular Weight:
183.21
Beilstein:
135859
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
97%
form
solid
mp
270-273 °C (lit.)
SMILES string
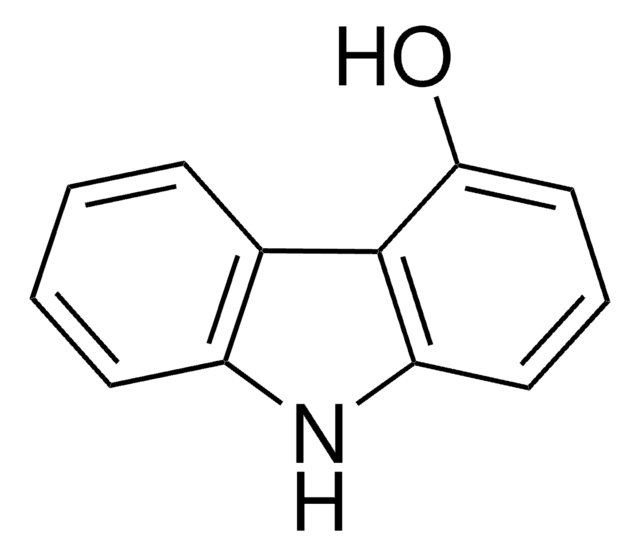
Oc1ccc2c(c1)[nH]c3ccccc23
InChI
1S/C12H9NO/c14-8-5-6-10-9-3-1-2-4-11(9)13-12(10)7-8/h1-7,13-14H
InChI key
GWPGDZPXOZATKL-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
2-Hydroxycarbazole is a compound structurally related to the Ca2+-mobilizing marine toxin, 9-methyl-7-bromoeudistomin. Room temperature electronic absorption and fluorescence spectra of 2-hydroxycarbazole has been studied in concentrated aqueous potassium hydroxide solutions. It undergoes chemoselective N-alkylation using NaH as a base in a THF-DMF solvent system.
Application
2-Hydroxycarbazole was used in the synthesis of isochromene fused carbazol, (4aS,13bR)-2,5,5-trimethyl-3,4,4a,5,8,13b-hexahydroisochromeno[3,4-b]carbazole.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Nguyen Manh Cuong et al.
Natural product communications, 4(7), 921-924 (2009-09-08)
The first synthesis of isochromene fused carbazols, (4aS, 13bR)-2,5,5-trimethyl-3,4,4a,5,8,13b-hexahydroisochromeno[3,4-b]carbazole (2) and its epi-isomer 3 by condensation of citral and 2-hydroxycarbazole using Ti(OEt)4 and MeAlC12 as catalysts is described.
Tamanna Mallick et al.
Colloids and surfaces. B, Biointerfaces, 172, 440-450 (2018-09-10)
Six structurally different carbazoles (1-6) were explored as the green reducing agents for the synthesis of the fluorescent Au nanoparticles with tailor-made morphology in anionic (sodium dodecyl sulphate, SDS), cationic (cetyltrimethylammonium bromide, CTAB) and neutral (polyvinylpyrrolidone, PVP) micelle medium. Structure
K Zawadzka et al.
Environmental science and pollution research international, 22(24), 19658-19666 (2015-08-16)
Nitrogen heterocyclic compounds, especially carbazole, quinolone, and pyridine are common types of environmental pollutants. Carbazole has a toxic influence on living organisms, and the knowledge of its persistence and bioconversion in ecosystems is still not complete. There is an increasing
S C Tovey et al.
European journal of pharmacology, 354(2-3), 245-251 (1998-10-01)
2-Hydroxycarbazole was shown to induce Ca2+ release from skeletal muscle and cardiac muscle sarcoplasmic reticulum at concentrations between 100-500 microM. This release was blocked by both 1 mM tetracaine and 30 microM ruthenium red which inhibit the ryanodine receptor or
Chemoselective N-alkylation of 2-hydroxycarbazole as a model for the synthesis of N-substituted pyrrole derivatives containing acidic functions.
Albanese D, et al.
Tetrahedron, 51(19), 5681-5688 (1995)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service