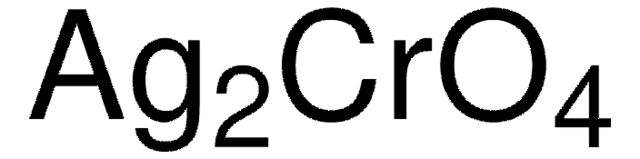00583
Nitrous oxide
≥99.998%
Synonym(s):
Dinitrogen monoxide, Laughing gas
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
N2O
CAS Number:
Molecular Weight:
44.01
Beilstein:
8137358
MDL number:
UNSPSC Code:
12142100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
vapor density
1.53 (15 °C, vs air)
vapor pressure
51.7 mmHg ( 21 °C)
Assay
≥99.998%
bp
−88 °C (lit.)
mp
−91 °C (lit.)
SMILES string
[O-][N+]#N
InChI
1S/N2O/c1-2-3
InChI key
GQPLMRYTRLFLPF-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
Nitrous oxide can be used as:
N2O can also be reacted with N-heterocyclic olefins (NHOs) to form electron-rich azo-bridged NHO dimers.
- A nitrogen donor to synthesize alkynyl and alkenyl substituted triazenes by coupling reaction with lithium amides and organomagnesium compounds.
- A reagent to synthesize azoimidazolium dyes.
N2O can also be reacted with N-heterocyclic olefins (NHOs) to form electron-rich azo-bridged NHO dimers.
Packaging
pressure tin
filling pressure at 15°C: 11 bar; content (15°C, 1 bar): 11 l
filling pressure at 15°C: 11 bar; content (15°C, 1 bar): 11 l
Other Notes
Sales restrictions may apply
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Ox. Gas 1 - Press. Gas Liquefied gas - STOT SE 3
Target Organs
Central nervous system
Storage Class Code
2A - Gases
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis of azoimidazolium dyes with nitrous oxide
Tskhovrebov AG, et al.
Angewandte Chemie (International Edition in English), 127, 1305-1308 (2015)
Synthesis of triazenes with nitrous oxide
Kiefer G, et al.
Angewandte Chemie (International Edition in English), 54, 302-305 (2015)
Synthesis of organic super-electron-donors by reaction of nitrous oxide with n-heterocyclic olefins
Eymann LYM, et al.
Journal of the American Chemical Society, 141, 17112-17116 (2019)
R L M Schils et al.
Animal : an international journal of animal bioscience, 7 Suppl 1, 29-40 (2012-10-04)
Herbivores are a significant source of nitrous oxide (N(2)O) emissions. They account for a large share of manure-related N(2)O emissions, as well as soil-related N(2)O emissions through the use of grazing land, and land for feed and forage production. It
Roberta Corona et al.
Fertility and sterility, 100(6), 1777-1783 (2013-10-12)
To evaluate the effect of addition of nitrous oxide (N2O) to the carbon dioxide (CO2) pneumoperitoneum (PP) and the effect of blood, plasma, or red blood cells (RBCs) on postoperative adhesions in a laparoscopic mouse model. Prospective randomized controlled trial.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service