392278
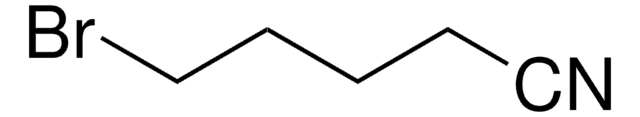
6-Bromohexanenitrile
95%
Synonym(s):
5-Bromopentyl cyanide, 6-Bromocapronitrile
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
Br(CH2)5CN
CAS Number:
Molecular Weight:
176.05
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
form
liquid
refractive index
n20/D 1.477 (lit.)
bp
134 °C/13 mmHg (lit.)
density
1.328 g/mL at 25 °C (lit.)
functional group
bromo
nitrile
SMILES string
BrCCCCCC#N
InChI
1S/C6H10BrN/c7-5-3-1-2-4-6-8/h1-5H2
InChI key
PHOSWLARCIBBJZ-UHFFFAOYSA-N
General description
6-Bromohexanenitrile (6-Bromohexanonitrile) is an ω-bromoalkanonitrile. Friedel Crafts alkylation of 6-bromohexanenitrile with benzene has been studied.
Application
6-Bromohexanenitrile (6-Bromocapronitrile, 6-Bromohexanonitrile) is suitable reagent used in the synthesis of (5-cyanopentyl)zinc(II) bromide, an organozinc reagent. It may be used in the synthesis of the following:
- 6-(1,4-dioxa-8-azaspiro[4.5]dec-8-yl)hexanenitrile.
- dimethyl 6,6′-dithiobiscaproimidate, a long-chain dithiobisimidate.
- N-benzyloxy-(4-cyanopentyl)-carbamic acid ethyl ester, a N-benzyloxy carbamate derivative.
- 1-hetarylsulfanyl-ω-cyanoalkanes.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Friedel-Crafts alkylation of benzene by normal ?-chloroalkanoic acids and their methyl esters and nitriles.
Zakharkin LI and Anikina EV
Bulletin of the Academy of Sciences of the USSR, Division of chemical science, 36(2), 327-330 (1987)
H Peretz et al.
European journal of biochemistry, 63(1), 77-82 (1976-03-16)
This communication describes a simple method for synthesizing cleavable bifunctional imido esters of different chain lengths. These reagents, which form covalent crosslinks between lysine residues of proteins, contain a disulfide bond which is cleaved under mild conditions by reducing agents
NICKEL-CATALYZED ENANTIOSELECTIVE NEGISHI CROSS-COUPLINGS OF RACEMIC SECONDARY α-BROMO AMIDES WITH ALKYLZINC REAGENTS: (S)-N-BENZYL-7-CYANO-2-ETHYL-N-PHENYLHEPTANAMIDE.
Sha Lou et al.
Organic syntheses; an annual publication of satisfactory methods for the preparation of organic chemicals, 87, 330-330 (2010-01-01)
?Green Chemical" Methods for the Regioselective Synthesis of 1-Hetarylsulfanyl-?-Cyanoalkanes."
Abele E, et al.
Latvijas Kimijas Zurnals, 49(1), 278-282 (2010)
Franziska Kupke et al.
Scientific reports, 6, 37631-37631 (2016-11-25)
Isothiocyanates are the most intensively studied breakdown products of glucosinolates from Brassica plants and well recognized for their pleiotropic effects against cancer but also for their genotoxic potential. However, knowledge about the bioactivity of glucosinolate-borne nitriles in foods is very
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service