All Photos(1)
About This Item
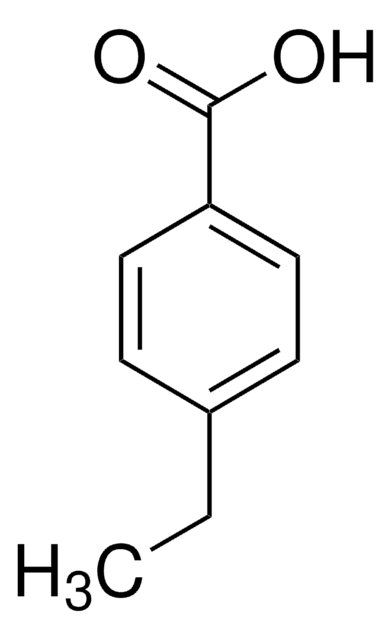
Linear Formula:
(CH3)3CC6H4CO2H
CAS Number:
Molecular Weight:
178.23
Beilstein:
607545
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
99%
mp
162-165 °C (lit.)
functional group
carboxylic acid
SMILES string
CC(C)(C)c1ccc(cc1)C(O)=O
InChI
1S/C11H14O2/c1-11(2,3)9-6-4-8(5-7-9)10(12)13/h4-7H,1-3H3,(H,12,13)
InChI key
KDVYCTOWXSLNNI-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
4-tert-Butylbenzoic acid was determined in water samples by means of liquid chromatography-electrospray ionisation mass spectrometry (LC-ESI-MS).
Application
4-tert-Butylbenzoic acid was used as a potent yeast sirtuin (Sir2p) inhibitor.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Acute Tox. 4 Oral - Aquatic Chronic 3 - Repr. 1B - STOT RE 1
Storage Class Code
6.1D - Non-combustible acute toxic Cat.3 / toxic hazardous materials or hazardous materials causing chronic effects
WGK
WGK 3
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Patrycja Makoś et al.
Journal of chromatography. A, 1517, 26-34 (2017-09-01)
The paper presents a new method for the determination of 15 carboxylic acids in samples of postoxidative effluents from the production of petroleum bitumens using ion-pair dispersive liquid-liquid microextraction and gas chromatography coupled to mass spectrometry with injection port derivatization.
Yi-Pei Chen et al.
Bioorganic & medicinal chemistry letters, 24(1), 349-352 (2013-11-26)
Employing a genetically modified yeast strain as a screening tool, 4-dimethylaminobenzoic acid (5) was isolated from the marine sediment-derived Streptomyces sp. CP27-53 as a weak yeast sirtuin (Sir2p) inhibitor. Using this compound as a scaffold, a series of disubstituted benzene
Adeline Martin et al.
Carbohydrate polymers, 93(2), 718-730 (2013-03-19)
The aim of this work was to develop the formation of multilayered coating incorporating a cyclodextrin polyelectrolyte onto a non-woven polyethylene terephthalate (PET) textile support in order to obtain reservoir and sustained release properties towards bioactive molecules. We optimized the
Inhibition of hepatic gluconeogenesis and lipogenesis by benzoic acid, p-tert.-butylbenzoic acid, and a structurally related hypolipidemic agent SC-33459.
S A McCune et al.
Archives of biochemistry and biophysics, 214(1), 124-133 (1982-03-01)
M D Whorton et al.
Scandinavian journal of work, environment & health, 7(3), 204-213 (1981-09-01)
The possible testicular effects resulting from occupational exposure to para-tertiary butyl benzoic acid (p-TBBA) were studied in 90 male volunteers employed at the Martinez, California, facility of the Shell Chemical Company. The comparison data used were obtained from an external
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service