M86804
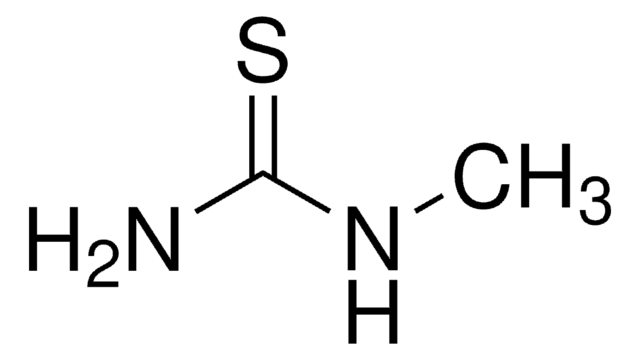
N-Methylurea
97%
Synonym(s):
1-Methylurea, N-Methylurea
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
CH3NHCONH2
CAS Number:
Molecular Weight:
74.08
Beilstein:
878189
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
97%
form
crystals
SMILES string
CNC(N)=O
InChI
1S/C2H6N2O/c1-4-2(3)5/h1H3,(H3,3,4,5)
InChI key
XGEGHDBEHXKFPX-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
J Guzmán Rincón et al.
Mutation research, 412(1), 69-81 (1998-03-21)
The in vivo nitrosation capacity of third-instar larvae of Drosophila melanogaster was assessed using the wing somatic mutation and recombination test (SMART). Larvate derived from two different crosses, the standard cross (ST) and the high bioactivation cross (HB) both involving
P J Groenen et al.
Food and chemical toxicology : an international journal published for the British Industrial Biological Research Association, 26(3), 215-225 (1988-03-01)
N-Nitroso compounds may well rank high among the genotoxic carcinogens present in our environment. Small amounts of such compounds may be formed in the human stomach after consumption of high-nitrate vegetables. Volatile nitrosamines can be conveniently determined but reliable methods
A J Streeter et al.
Drug metabolism and disposition: the biological fate of chemicals, 18(4), 447-452 (1990-07-01)
The single-dose toxicokinetics of monomethylamine has been characterized in the rat by HPLC assay of serial blood samples. Biphasic first-order elimination was observed following an iv bolus dose of 19 mumol/kg with a terminal half-life of 19.1 +/- 1.3 min
L S Povarov et al.
Bioorganicheskaia khimiia, 16(4), 559-568 (1990-04-01)
Derivatives of antitumour anthracycline antibiotics containing N-methylurea moiety in the carbohydrate ring were obtained by the interaction of methyl isocyanate with daunorubicin, doxorubicin, carminomycin and daunorubicin derivatives, substituted at C-13 or C-14 positions. N-Nitrosation of these compounds yielded modified anthracycline
V V Matveev
Tsitologiia, 31(12), 1459-1465 (1989-12-01)
Following short-term local applications of high doses of N-methyl-N-nitrozourea and 7,2-dimethylbenz(a)antracen, the intercellular adhesion in the distal colon epithelium of ice retains increased at least for a month. The magnitude of this shift and its duration ops both with moving
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service