115819
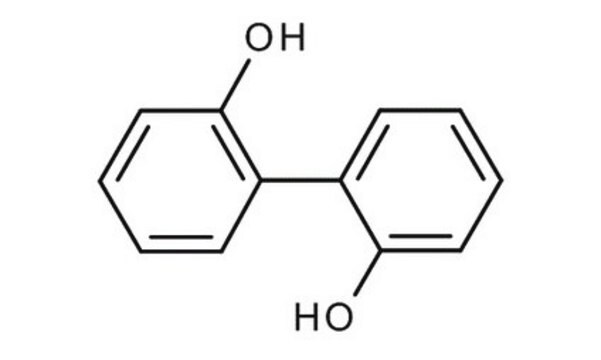
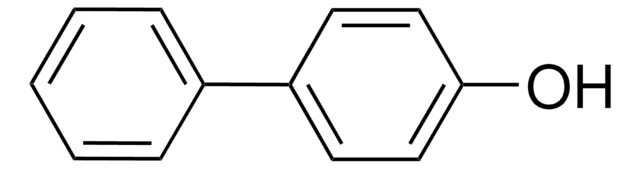
2,2′-Biphenol
99%
Synonym(s):
2,2′-Biphenyldiol, 2,2′-Dihydroxybiphenyl, 2,2′-Diphenol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
HOC6H4C6H4OH
CAS Number:
Molecular Weight:
186.21
Beilstein:
1638363
EC Number:
MDL number:
UNSPSC Code:
12162002
PubChem Substance ID:
NACRES:
NA.23
Recommended Products
Quality Level
Assay
99%
form
solid
bp
315 °C (lit.)
mp
108-110 °C (lit.)
SMILES string
Oc1ccccc1-c2ccccc2O
InChI
1S/C12H10O2/c13-11-7-3-1-5-9(11)10-6-2-4-8-12(10)14/h1-8,13-14H
InChI key
IMHDGJOMLMDPJN-UHFFFAOYSA-N
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
309.2 °F - closed cup - (External MSDS)
Flash Point(C)
154 °C - closed cup - (External MSDS)
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Sierra Rayne et al.
Photochemical & photobiological sciences : Official journal of the European Photochemistry Association and the European Society for Photobiology, 4(11), 876-886 (2005-10-28)
Photochemical studies on a range of model dibenzo[1,4]dioxins were performed in aqueous and organic solutions. The compounds were found to undergo a photochemically initiated aryl-ether bond homolysis that yields reactive 2-spiro-6'-cyclohexa-2',4'-dien-1'-one and subsequent 2,2'-biphenylquinone intermediates. Under steady-state irradiation, the 2,2'-biphenylquinones
Christina DiMarco-Crook et al.
Journal of food science, 85(4), 1292-1301 (2020-03-08)
Chemoprevention strategies employing the use of multiple dietary bioactive components and their metabolites in combination offer advantages due to their low toxicity and potential synergistic interactions. Herein, for the first time, we studied the combination of curcumin and 3',4'-didemethylnobiletin (DDMN)
Bernd Schmidt et al.
The Journal of organic chemistry, 78(17), 8680-8688 (2013-08-01)
User-friendly protocols for the protecting group-free synthesis of 2,2'-biphenols via Suzuki-Miyaura coupling of o-halophenols and o-boronophenol are presented. The reactions proceed in water in the presence of simple additives such as K2CO3, KOH, KF, or TBAF and with commercially available
R J Heath et al.
The Journal of biological chemistry, 273(46), 30316-30320 (1998-11-07)
The broad spectrum antibacterial properties of 2-hydroxydiphenyl ethers have been appreciated for decades, and their use in consumer products is rapidly increasing. We identify the enoyl-acyl carrier protein reductase (fabI) component of the type II fatty acid synthase system as
W A Prütz et al.
International journal of radiation biology and related studies in physics, chemistry, and medicine, 44(2), 183-196 (1983-08-01)
Phenoxyl radicals generated pulse radiolytically by the reaction of N.3 with Gly-Tyr decay biomolecularly (2k = 4.7 X 10(8)M-1 s-1) with efficient formation of 2,2'-dimers, which enolize rapidly (k = 2.7 X 10(4) s-1) to produce the 2,2'-biphenolic product. The
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service