L1876
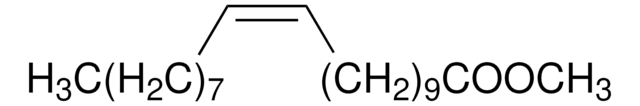
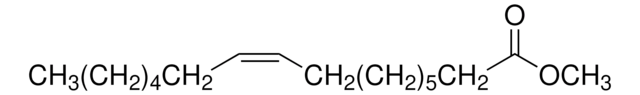
Methyl linoleate
≥98% (GC)
Sinónimos:
Linoleic acid methyl ester, Methyl cis,cis-9,12-octadecadienoate
About This Item
Productos recomendados
Nivel de calidad
Ensayo
≥98% (GC)
Formulario
liquid
índice de refracción
n20/D 1.462 (lit.)
bp
192 °C/4 mmHg (lit.)
mp
−35 °C (lit.)
densidad
0.889 g/mL at 25 °C (lit.)
grupo funcional
ester
tipo de lípido
omega FAs
Condiciones de envío
ambient
temp. de almacenamiento
−20°C
cadena SMILES
CCCCC\C=C/C\C=C/CCCCCCCC(=O)OC
InChI
1S/C19H34O2/c1-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-18-19(20)21-2/h7-8,10-11H,3-6,9,12-18H2,1-2H3/b8-7-,11-10-
Clave InChI
WTTJVINHCBCLGX-NQLNTKRDSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Descripción general
Aplicación
Acciones bioquímicas o fisiológicas
Código de clase de almacenamiento
10 - Combustible liquids
Clase de riesgo para el agua (WGK)
WGK 1
Equipo de protección personal
Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico