8.52418
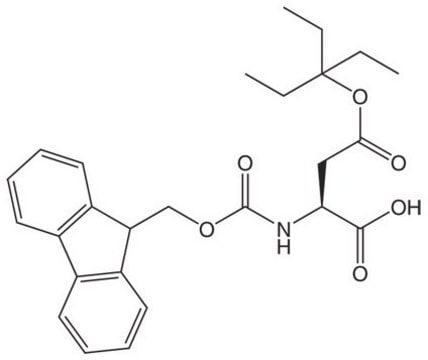
Fmoc-Asp(OBno)-OH
for peptide synthesis, Novabiochem®
Sinónimos:
Fmoc-Asp(OBno)-OH
About This Item
Productos recomendados
product name
Fmoc-Asp(OBno)-OH, Novabiochem®
Quality Level
product line
Novabiochem®
form
powder
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
manufacturer/tradename
Novabiochem®
application(s)
peptide synthesis
functional group
carboxylic acid
storage temp.
−20°C (−15°C to −25°C)
General description
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Overcoming Aspartimide Formation in Fmoc SPPS
Literature references:
[1] R. Behrendt, et al. (2015) J. Pept. Sci., 21, 680.
[2] R. Behrendt, et al. (2016) J. Pept. Sci., 22, 92.
Application
Analysis Note
Appearance of substance (visual): powder, chunks or crystals
Identity (IR): passes test
Purity (TLC (018A)): ≥ 95 %
Enantiomeric purity: ≥ 99.5 % (a/a)
Assay (HPLC, area%): ≥ 97.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
To see the solvent systems used for TLC of Novabiochem® products please click here.
Legal Information
Storage Class
11 - Combustible Solids
wgk_germany
WGK 2
flash_point_f
Not applicable
flash_point_c
Not applicable
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Artículos
Aspartimide formation 1,2 is caused by repeated exposure of aspartic acid-containing sequences to bases like piperidine and can result ultimately in the generation of 9 different by-products.
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico