307556
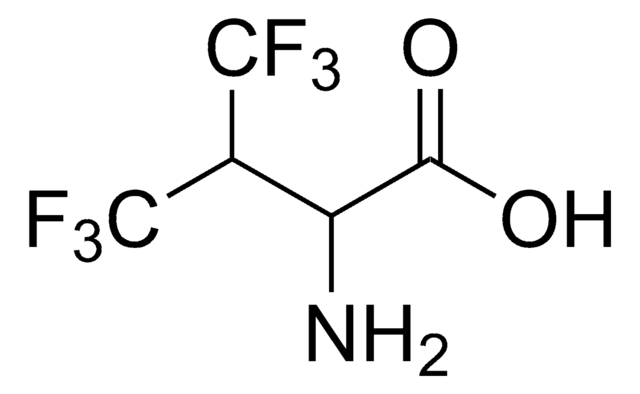
3,3,3-Trifluoro-DL-alanine
98%
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula lineal:
CF3CH(NH2)CO2H
Número de CAS:
Peso molecular:
143.06
EC Number:
MDL number:
UNSPSC Code:
12352209
PubChem Substance ID:
NACRES:
NA.22
Productos recomendados
assay
98%
reaction suitability
reaction type: solution phase peptide synthesis
mp
231-234 °C (lit.)
application(s)
peptide synthesis
storage temp.
2-8°C
SMILES string
NC(C(O)=O)C(F)(F)F
InChI
1S/C3H4F3NO2/c4-3(5,6)1(7)2(8)9/h1H,7H2,(H,8,9)
InChI key
HMJQKIDUCWWIBW-UHFFFAOYSA-N
Storage Class
11 - Combustible Solids
wgk_germany
WGK 3
ppe
dust mask type N95 (US), Eyeshields, Gloves
Certificados de análisis (COA)
Busque Certificados de análisis (COA) introduciendo el número de lote del producto. Los números de lote se encuentran en la etiqueta del producto después de las palabras «Lot» o «Batch»
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
L Pollegioni et al.
FEBS letters, 507(3), 323-326 (2001-11-07)
D-Amino acid oxidase (DAAO) is a flavoprotein oxidase that catalyzes the oxidation of amino acids and produces ketoacids and H(2)O(2). The rate of product release from reduced DAAO from Rhodotorula gracilis is pH dependent and reflects a pK(a) of approximately
T Clausen et al.
Journal of molecular biology, 262(2), 202-224 (1996-09-20)
Cystathionine beta-lyase (CBL) is a member of the gamma-family of PLP-dependent enzymes, that cleaves C beta-S bonds of a broad variety of substrates. The crystal structure of CBL from E. coli has been solved using MIR phases in combination with
Yong Guo et al.
Organic letters, 8(5), 827-829 (2006-02-24)
2-Aminoperfluoropropene has been prepared by the Mg-promoted defluorinative N-silylation of N-p-methoxyphenyl hexafluoroacetone imine and has been employed as a synthon of trifluoroalanine for the preparation of trifluoroalanine dipeptides.
C W Fearon et al.
Biochemistry, 21(16), 3790-3794 (1982-08-03)
Inactivation of gamma-cystathionase by beta, beta, beta-trifluoroalanine, a suicide inactivator of the enzyme, results in covalent labeling of an amino group of the protein [Silverman, R. B., & Abeles, R. H. (1977) Biochemistry 16, 5515-5520]. We have established that this
R S Phillips et al.
Archives of biochemistry and biophysics, 296(2), 489-496 (1992-08-01)
Trifluoroalanine is a mechanism-based inactivator of Escherichia coli tryptophan indole-lyase (tryptophanase) and E. coli tryptophan synthase (R. B. Silverman and R. H. Abeles, 1976, Biochemistry 15, 4718-4723). We have found that indole is able to prevent inactivation of tryptophan indole-lyase
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico