304484
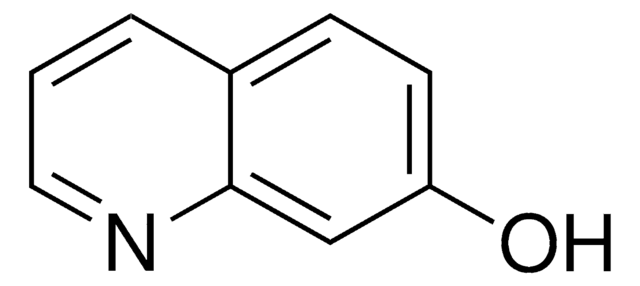
6-Hydroxyquinoline
95%
Sinónimos:
6-Quinolinol, 6-Hydroxyquinoline
Iniciar sesiónpara Ver la Fijación de precios por contrato y de la organización
About This Item
Fórmula empírica (notación de Hill):
C9H7NO
Número de CAS:
Peso molecular:
145.16
Beilstein:
113196
Número CE:
Número MDL:
Código UNSPSC:
12352100
ID de la sustancia en PubChem:
NACRES:
NA.22
Productos recomendados
Nivel de calidad
Ensayo
95%
mp
188-190 °C (lit.)
cadena SMILES
Oc1ccc2ncccc2c1
InChI
1S/C9H7NO/c11-8-3-4-9-7(6-8)2-1-5-10-9/h1-6,11H
Clave InChI
OVYWMEWYEJLIER-UHFFFAOYSA-N
¿Está buscando productos similares? Visita Guía de comparación de productos
Categorías relacionadas
Descripción general
6-Hydroxyquinoline is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. The excited-state proton transfer and geminate recombination of 6-hydroxyquinoline encaged in catalytic Na+-exchanged faujasite zeolites X and Y have been explored by measuring steady-state and picosecond time-resolved spectra.
Aplicación
6-Hydroxyquinoline was used in synthesis of 2,6-substituted-benzo[d]thiazole analogs and 2,4-substituted-benzo[d]thiazole analogs.
Palabra de señalización
Warning
Frases de peligro
Consejos de prudencia
Clasificaciones de peligro
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Órganos de actuación
Respiratory system
Código de clase de almacenamiento
11 - Combustible Solids
Clase de riesgo para el agua (WGK)
WGK 3
Punto de inflamabilidad (°F)
Not applicable
Punto de inflamabilidad (°C)
Not applicable
Equipo de protección personal
dust mask type N95 (US), Eyeshields, Gloves
Elija entre una de las versiones más recientes:
¿Ya tiene este producto?
Encuentre la documentación para los productos que ha comprado recientemente en la Biblioteca de documentos.
Los clientes también vieron
Yu-Hui Liu et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 128, 280-284 (2014-04-01)
6-Hydroxyquinoline (6HQ) is an ideal photoacid system for exploring excited-state proton transfer (ESPT) reactions. We have previously (Mahata et al. (2002)) shown that the ESPT reaction between 6HQ and trimethylamine (TMA) leads to an "unusual" emission in the 440-450 nm
Katharigatta Narayanaswamy Venugopala et al.
European journal of medicinal chemistry, 65, 295-303 (2013-06-04)
A novel and efficient one pot synthesis was developed for 2,6-substituted-benzo[d]thiazole analogues 4a-k and 2,4-substituted-benzo[d]thiazole analogues 4l-pvia three component condensation reaction of substituted arylaldehyde, 2-amino-6-halo/4-methyl-benzo[d]thiazole and 2-naphthol or 6-hydroxyquinoline in presence of 10% w/v NaCl in water by microwave method.
M S Mehata et al.
Spectrochimica acta. Part A, Molecular and biomolecular spectroscopy, 59(3), 559-567 (2003-01-14)
Fluorescence characteristics of 6-hydroxyquinoline (6-HQ) have been studied at room temperature in Nafion(R) film by steady state and nano-second time-resolved fluorescence spectroscopy. The fluorescence spectrum exhibits single emission band corresponding to the protonated form of 6-HQ in this matrix. However
Sun-Young Park et al.
Chemistry (Weinheim an der Bergstrasse, Germany), 16(42), 12609-12615 (2010-09-21)
The excited-state proton transfer and geminate recombination of 6-hydroxyquinoline (6HQ) encaged in catalytic Na(+)-exchanged faujasite zeolites X (NaX) and Y (NaY) have been explored by measuring steady-state and picosecond time-resolved spectra. The pathways and rate constants of proton transfer of
Mohan Singh Mehata
The journal of physical chemistry. B, 112(28), 8383-8386 (2008-06-20)
A hydrogen-bonded network formed between 6-hydroxyquinoline (6-HQ) and acetic acid (AcOH) has been characterized using a time-resolved fluorescence technique. In the bridged hydrogen-bonded complex of cis-6-HQ and AcOH, an excited-state reaction proceeds via proton transfer along the hydrogen bond, resulting
Nuestro equipo de científicos tiene experiencia en todas las áreas de investigación: Ciencias de la vida, Ciencia de los materiales, Síntesis química, Cromatografía, Analítica y muchas otras.
Póngase en contacto con el Servicio técnico