638080
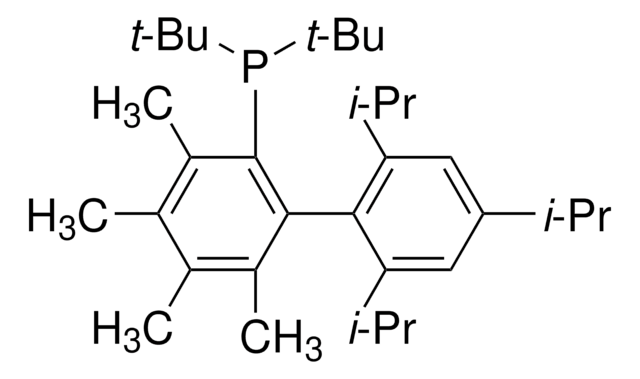
tBuXPhos
98%
Synonym(s):
tBuXPhos, 2-Di-tert-butylphosphino-2′,4′,6′-triisopropylbiphenyl, t-Bu XPhos, tert-Butyl XPhos
About This Item
Recommended Products
Quality Level
Assay
98%
form
solid
reaction suitability
reaction type: Cross Couplings
reagent type: ligand
reaction type: Arylations
reagent type: ligand
reaction type: Buchwald-Hartwig Cross Coupling Reaction
reagent type: ligand
reaction type: Carboxylations
reagent type: ligand
reaction type: Decarboxylations
greener alternative product score
old score: 12
new score: 1
Find out more about DOZN™ Scoring
greener alternative product characteristics
Atom Economy
Design for Energy Efficiency
Use of Renewable Feedstocks
Learn more about the Principles of Green Chemistry.
sustainability
Greener Alternative Product
mp
148-151 °C (lit.)
functional group
phosphine
greener alternative category
SMILES string
CC(P(C(C=CC=C1)=C1C(C(C(C)C)=CC(C(C)C)=C2)=C2C(C)C)C(C)(C)C)(C)C
InChI
1S/C29H45P/c1-19(2)22-17-24(20(3)4)27(25(18-22)21(5)6)23-15-13-14-16-26(23)30(28(7,8)9)29(10,11)12/h13-21H,1-12H3
InChI key
SACNIGZYDTUHKB-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Learn more about Buchwald Phosphine Ligands
Application
It can be used in the following reactions:
- Palladium-catalyzed Tsuji-Trost substitution and cross-coupling of benzylic fluorides.
- Palladium-catalyzed C-N cross-coupling of sulfinamides and aryl halides.
- Palladium-catalyzed rapid methoxylation and deuteriomethoxylation of bromo-chalcones.
Legal Information
related product
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Buchwald Phosphine Ligands
Buchwald phosphine ligands for C-C, C-N, and C-O bond formation.
Related Content
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Explore reliable, premium grade catalysis materials for your pharma or industrial project. Specialty chemicals and formulations are available in bulk quantities and volumes from a few grams to multi-metric tons with complete documentation to simplify your leap from development to commercialization.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service