All Photos(1)
About This Item
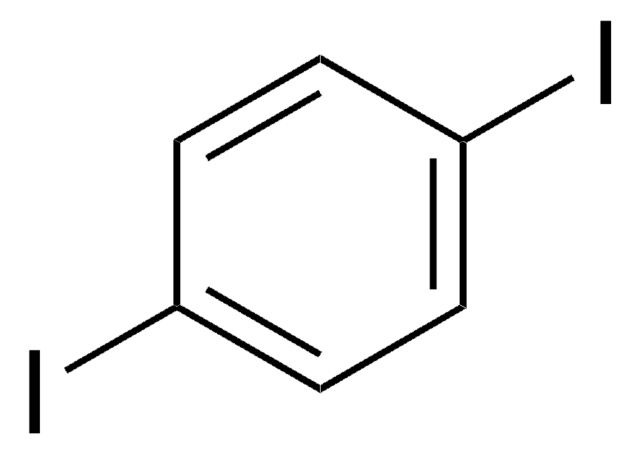
Empirical Formula (Hill Notation):
C6H4I2
CAS Number:
Molecular Weight:
329.90
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
Assay
98%
storage condition
protect from light
mp
34-37 °C (lit.)
storage temp.
2-8°C
SMILES string
Ic1cccc(I)c1
InChI
1S/C6H4I2/c7-5-2-1-3-6(8)4-5/h1-4H
InChI key
SFPQFQUXAJOWNF-UHFFFAOYSA-N
General description
1,3-Diiodobenzene is a halogenated benzene derivative. Its reaction with phenylboronic acid in the presence of CuI, DABCO (1,4-diazabicyclo[2.2.2]octane) and TBAB (n-Bu4NBr) has been analyzed. 1,3-Diiodobenzene undergoes coupling with 2-methylthiophene in the presence of Ir/Ag2CO3 to afford meta-linked isomer of thiophene-benzene-thiophene triad.
Application
1,3-Diiodobenzene may be used in the synthesis of:
- 3,5-bis(perfluorodecyl)phenylboronic acid
- epitaxially aligned and separated polyphenylene lines on Cu(110)
- 1,3-bis(4-ethynyl-2,5-dibutoxyphenyl-1-ethynyl)benzene
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
235.4 °F - closed cup
Flash Point(C)
113 °C - closed cup
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Synthesis, Chain Rigidity, and Luminescent Properties of Poly [(1, 3-phenyleneethynylene)-a lt-tris (2, 5-dialkoxy-1, 4-phenyleneethynylene)] s.
Chu Q, et al.
Macromolecules, 35(20), 7569-7574 (2002)
3, 5-Bis (perfluorodecyl) phenylboronic acid as an easily recyclable direct amide condensation catalyst.
Ishihara K, et al.
Synlett, 2001(09), 1371-1374 (2001)
Jin-Heng Li et al.
The Journal of organic chemistry, 72(6), 2053-2057 (2007-02-09)
In the presence of TBAB, CuI-catalyzed Suzuki-Miyaura cross-coupling of vinyl halides and aryl halides with arylboronic acids was conducted smoothly to afford the corresponding diarylethenes and polyaryls in moderate to good yields using DABCO (1,4-diazabicyclo[2.2.2]octane) as the ligand. We also
Benoît Join et al.
Angewandte Chemie (International ed. in English), 48(20), 3644-3647 (2009-04-09)
Efficient couplings using equimolar quantities of each coupling partner and multiple C-H bond arylation reactions are achieved with an Ir-based catalytic system for the C-H bond arylation of electron-rich heteroarenes with iodoarenes to construct extended pi-systems. The dramatic ligand effect
J A Lipton-Duffin et al.
Small (Weinheim an der Bergstrasse, Germany), 5(5), 592-597 (2009-02-26)
The surface-mediated synthesis of epitaxially aligned and separated polyphenylene lines on Cu(110) by exploiting the Ullmann dehalogenation reaction is reported. Scanning tunneling microscopy (STM) and X-ray photoelectron spectroscopy (XPS) show that the C-I bonds of 1,4-diiodobenzene and 1,3-diiodobenzene (C(6)H(4)I(2)) are
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service