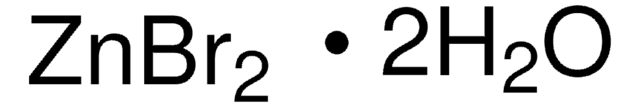223883
Zinc iodide
≥98%
Synonym(s):
Diiodozinc, Zinc diiodide
About This Item
Recommended Products
Assay
≥98%
form
powder
reaction suitability
reagent type: catalyst
core: zinc
mp
445 °C (lit.)
density
4.74 g/mL at 25 °C (lit.)
SMILES string
I[Zn]I
InChI
1S/2HI.Zn/h2*1H;/q;;+2/p-2
InChI key
UAYWVJHJZHQCIE-UHFFFAOYSA-L
Looking for similar products? Visit Product Comparison Guide
Related Categories
Application
- As an activator to synthesize poly(vinyl methyl ether) via living cationic polymerization. ZnI2 is known to produce living vinyl ether polymerizations.
- To prepare precursor solutions for lead halide perovskite solar cells. The addition of ZnI2 leads to higher stability against environmental moisture when introduced into perovskite cells.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Acute 1 - Aquatic Chronic 1 - Eye Irrit. 2 - Skin Irrit. 2 - STOT RE 2 Oral
Target Organs
Thyroid
Storage Class Code
13 - Non Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Articles
Colloidal quantum dots (CQDs) are semiconducting crystals of only a few nanometers (ca. 2–12 nm) coated with ligand/surfactant molecules to help prevent agglomeration.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service