498491
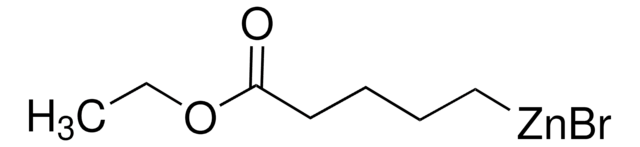
4-Ethoxy-4-oxobutylzinc bromide solution
0.5 M in THF
Synonym(s):
(4-Ethoxy-4-oxobutyl)zinc(II) bromide, Bromo[4-ethoxy-4-(oxo-κO)butyl-κC]zinc
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
C2H5O2C(CH2)3ZnBr
CAS Number:
Molecular Weight:
260.44
MDL number:
UNSPSC Code:
12352103
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Quality Level
concentration
0.5 M in THF
density
0.976 g/mL at 25 °C
functional group
ester
storage temp.
2-8°C
SMILES string
CCOC(=O)CCC[Zn]Br
InChI
1S/C6H11O2.BrH.Zn/c1-3-5-6(7)8-4-2;;/h1,3-5H2,2H3;1H;/q;;+1/p-1
InChI key
QRIVZBPOANGBFR-UHFFFAOYSA-M
Related Categories
Application
4-Ethoxy-4-oxobutylzinc bromide can be used:
- As an intermediate in one of the key synthetic steps for the preparation of acrylic acid derivatives as EP3 receptor antagonists.
- As an intermediate in the final step of the total synthesis of a natural product mucosin.
- As a substrate in the synthesis of multi-substituted haloalkenes by reacting with 1-haloalkenes via Negishi cross-coupling reaction.
Legal Information
Product of Rieke Metals, Inc.
Rieke is a registered trademark of Rieke Metals, Inc.
Rieke is a registered trademark of Rieke Metals, Inc.
recommended
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Carc. 2 - Eye Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Supplementary Hazards
Storage Class Code
3 - Flammable liquids
WGK
WGK 3
Flash Point(F)
-6.2 °F - (THF)
Flash Point(C)
-21.2 °C - (THF)
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Discovery of a series of acrylic acids and their derivatives as chemical leads for selective EP3 receptor antagonists
Asada M, et al.
Bioorganic & Medicinal Chemistry, 17(18), 6567-6582 (2009)
Stereopermutation on the Putative Structure of the Marine Natural Product Mucosin
Antonsen SG, et al.
Molecules (Basel), 22(10), 1720-1720 (2017)
Total Synthesis of (-)-Mucosin and Revision of Structure
Nolsoe JMJ, et al.
The Journal of Organic Chemistry, 83(24), 15066-15076 (2018)
Synthesis of the multisubstituted halogenated olefins via cross-coupling of dihaloalkenes with alkylzinc bromides
Andrei D and Wnuk SF
The Journal of Organic Chemistry, 71(1), 405-408 (2006)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service