C3934
Carbonic Anhydrase from bovine erythrocytes
lyophilized powder, ≥2,000 W-A units/mg protein
Synonym(s):
Carbonate Dehydratase, Carbonate Hydrolyase
About This Item
Recommended Products
biological source
bovine erythrocytes
form
lyophilized powder
specific activity
≥2,000 W-A units/mg protein
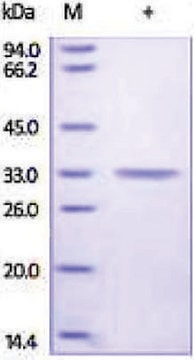
mol wt
30 kDa
application(s)
diagnostic assay manufacturing
storage temp.
2-8°C
Looking for similar products? Visit Product Comparison Guide
Application
Biochem/physiol Actions
Unit Definition
inhibitor
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Resp. Sens. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Protocols
Objective: To standardize a procedure for the enzymatic assay of Carbonic Anhydrase (EC 4.2.1.1) for Wilbur-Anderson Units.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service