8.52107
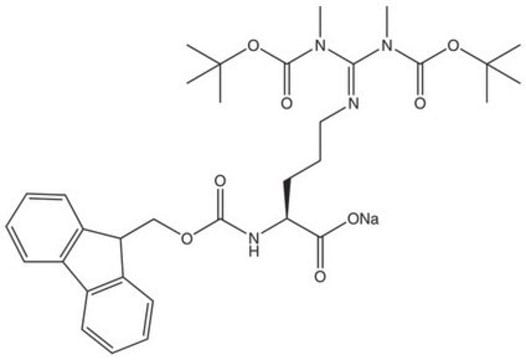
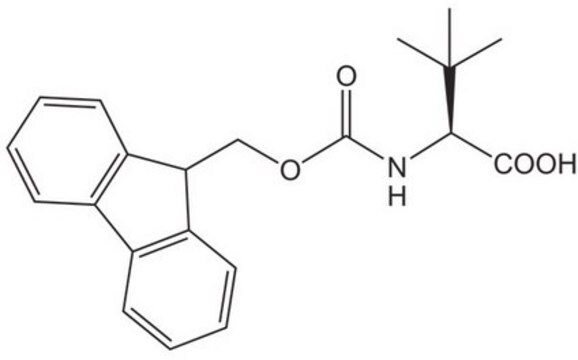
Fmoc-ADMA(Pbf)-OH
≥98% (TLC), for peptide synthesis, Novabiochem®
Synonym(s):
Fmoc-ADMA(Pbf)-OH, N-α-Fmoc-N,N-ω-dimethyl-N-ωÆ-(2,2,4,6,7-pentamethyldihydrobenzofuran-5-sulfonyl)-L-arginine
About This Item
Recommended Products
product name
Fmoc-ADMA(Pbf)-OH, Novabiochem®
Quality Level
product line
Novabiochem®
Assay
≥96.0% (HPLC)
≥98% (TLC)
form
powder
reaction suitability
reaction type: Fmoc solid-phase peptide synthesis
manufacturer/tradename
Novabiochem®
application(s)
peptide synthesis
functional group
Fmoc
storage temp.
15-25°C
General description
Associated Protocols and Technical Articles
Cleavage and Deprotection Protocols for Fmoc SPPS
Literature references
[1] S. Rothbart, et al. (2012) Methods Enzymol., 512, 107.
Application
- Effect of arginine methylation on the RNA recognition and cellular uptake of Tat-derived peptides: Studies the use of Fmoc-ADMA(Pbf)-OH in modifying peptides to enhance their cellular uptake, relevant for drug delivery systems and therapeutic molecule development (JH Li et al., 2015).
- Advances in Fmoc solid-phase peptide synthesis: Reviews the role of Fmoc-ADMA(Pbf)-OH in advancing peptide synthesis techniques, impacting the field of material science through the development of novel synthetic strategies and applications in biomaterials (R Behrendt, J Offer, 2016).
Linkage
Analysis Note
Appearance of substance (visual): powder
Identity (IR): passes test
Purity (TLC(157A)): ≥ 98 %
Enantiomeric purity: ≥ 99.0 % (a/a)
Assay (HPLC, area%): ≥ 96.0 % (a/a)
Solubility (1 mmole in 2 ml DMF): clearly soluble
Water (K. F.): ≤ 2.0 %
To see the solvent systems used for TLC of Novabiochem® products please click here.
Legal Information
Storage Class Code
11 - Combustible Solids
WGK
WGK 2
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Articles
Unnatural amino acids, the non-proteinogenic amino acids that either occur naturally or are chemically synthesized, are becoming more and more important as tools for modern drug discovery research.
Protocols
We provide an overview of our available reagents, together with recommendations and details of their use for synthesis of peptides containing post-translationally modified amino acids.
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service