P56100
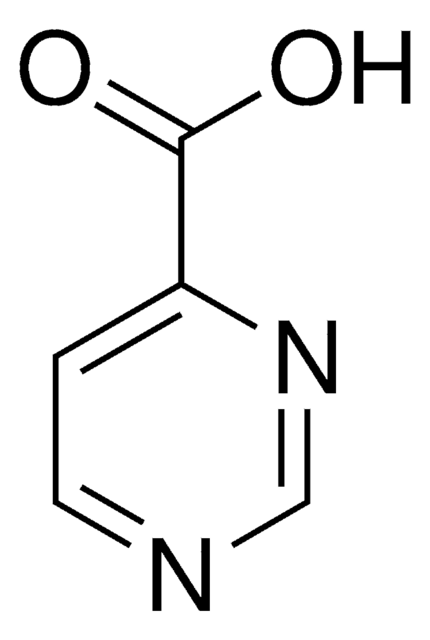
Pyrazinecarboxylic acid
99%
Synonym(s):
Pyrazinoic acid
Sign Into View Organizational & Contract Pricing
All Photos(2)
About This Item
Empirical Formula (Hill Notation):
C5H4N2O2
CAS Number:
Molecular Weight:
124.10
Beilstein:
112305
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
form
powder
mp
222-225 °C (dec.) (lit.)
SMILES string
OC(=O)c1cnccn1
InChI
1S/C5H4N2O2/c8-5(9)4-3-6-1-2-7-4/h1-3H,(H,8,9)
InChI key
NIPZZXUFJPQHNH-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Mohamed Abdel-Aziz et al.
European journal of medicinal chemistry, 45(8), 3384-3388 (2010-05-22)
A series of pyrazine-2-carboxylic acid hydrazide derivatives were synthesized and screened for their activity against Mycobacterium tuberculosis. The results show that pyrazine-2-carboxylic acid hydrazide-hydrazone derivatives 3a-l were less active than pyrazinamide. In contrast, the N(4)-ethyl-N(1)-pyrazinoyl-thiosemicarbazide 4 showed the highest activity
Wolfgang Holzer et al.
Magnetic resonance in chemistry : MRC, 47(7), 617-624 (2009-04-30)
NMR spectroscopic studies are undertaken with derivatives of 2-pyrazinecarboxylic acid. Complete and unambiguous assignment of chemical shifts ((1)H, (13)C, (15)N) and coupling constants ((1)H,(1)H; (13)C,(1)H; (15)N,(1)H) is achieved by combined application of various 1D and 2D NMR spectroscopic techniques. Unequivocal
Mirko Zimic et al.
Tuberculosis (Edinburgh, Scotland), 92(1), 84-91 (2011-10-19)
Pyrazinamide is one of the most important drugs in the treatment of latent Mycobacterium tuberculosis infection. The emergence of strains resistant to pyrazinamide represents an important public health problem, as both first- and second-line treatment regimens include pyrazinamide. The accepted
Stefania Tanase et al.
Dalton transactions (Cambridge, England : 2003), (15)(15), 2026-2033 (2008-04-03)
The reactivity towards H(2)O(2) of the complexes [Fe(pca)(2)(py)(2)].py (1) and Na(2){[Fe(pca(3))](2)O}.2H(2)O.CH(3)CN (2) (where pca(-) is pyrazine-2-carboxylate) and their catalytic activity in the oxidation of hydrocarbons is reported. Addition of H(2)O(2) to 1 results in the formation of a dinuclear Fe(III)-(mu-O)-Fe(III)
Ping Lu et al.
Antimicrobial agents and chemotherapy, 55(11), 5354-5357 (2011-08-31)
Pyrazinoic acid, the active form of the first-line antituberculosis drug pyrazinamide, decreased the proton motive force and respiratory ATP synthesis rates in subcellular mycobacterial membrane assays. Pyrazinoic acid also significantly lowered cellular ATP levels in Mycobacterium bovis BCG. These results
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service