All Photos(1)
About This Item
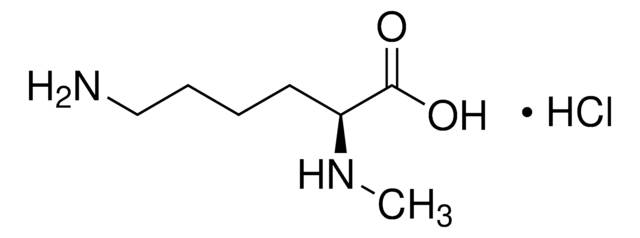
Empirical Formula (Hill Notation):
C7H16N2O2 · HCl
CAS Number:
Molecular Weight:
196.68
EC Number:
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
NACRES:
NA.32
grade:
for analytical purposes
Recommended Products
grade
for analytical purposes
Quality Level
Assay
≥98.0% (TLC)
optical activity
[α]/D 20.5±1.5°, c = 0.1 in 1 M HCl
storage temp.
2-8°C
SMILES string
Cl.CNCCCC[C@H](N)C(O)=O
Cl.CNCCCC[C@H](N)C(O)=O
InChI
1S/C7H16N2O2.ClH/c1-9-5-3-2-4-6(8)7(10)11;/h6,9H,2-5,8H2,1H3,(H,10,11);1H/t6-;/m0./s1
InChI key
AQELUQTVJOFFBN-RGMNGODLSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
N ε-methyl-L-lysine was identified as a lysine analog with inhibitory effects on the growth and sporulation of Penicillium chrysogenum and benzyl-penicillin formation by mycelia.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
C G Friedrich et al.
Applied and environmental microbiology, 34(6), 706-709 (1977-12-01)
Compounds structurally related to lysine were tested against Penicillium chrysogenum Wis. 54-1255 for inhibition of growth, sporulation, and penicillin formation. This strain is relatively resistant to lysine analogs. The compounds that were the more active inhibitors of growth and whose
M Friedman et al.
The Journal of nutrition, 111(8), 1362-1369 (1981-08-01)
Growth assays using mice on synthetic amino acid diets showed that substituting epsilon-N-methyl-L-lysine, epsilon-N-dimethyl-L-lysine and epsilon-N-trimethyl-L-lysine for lysine resulted in relative replacement values about 1/12, 1/20 and 1/25, respectively, of that obtained with the standard lysine diet. Similar studies showed
H Kalász et al.
Journal of chromatographic science, 43(4), 165-168 (2005-06-25)
Administration of (14)C-labelled L-deprenyl to rats results in the urinary elimination of a 14C-labelled compound. The 9-fluorenylmethoxycarbonyl chloride-reacted urine sample is fractionated by high-performance liquid chromatography (HPLC) on an octadecyl silica stationary phase. N(epsilon)-Monomethyl-lysine is identified in the fraction containing
H Kalász et al.
Journal of chromatography. A, 1079(1-2), 208-212 (2005-07-26)
Nepsilon-Monomethyllysine was identified in the serum, urine, brain, and liver samples of rats treated per os with L-deprenyl. The identification procedure included reaction with Fmoc chloride, clean-up, and analysis using HPLC-UV-MS. Oral administration of (-)-N-14C-methyl-N-propynyl(2-phenyl-1-methyl)ethylammonium hydrochloride L-deprenyl) to rats resulted
M Moracci et al.
Enzyme and microbial technology, 17(11), 992-997 (1995-11-01)
The gene coding for the beta-glycosidase from the archaeon Sulfolobus solfataricus has been overexpressed in Escherichia coli. The enzyme was purified to homogeneity with a rapid purification procedure employing a thermal precipitation as a crucial step. The final yield was
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service