About This Item
推荐产品
化驗
97%
形狀
liquid
反應適用性
reaction type: C-C Bond Formation
reagent type: catalyst
reaction type: C-H Activation
折射率
n20/D 1.431 (lit.)
n20/D 1.432
bp
186 °C (lit.)
密度
1.662 g/mL at 25 °C
1.687 g/mL at 25 °C (lit.)
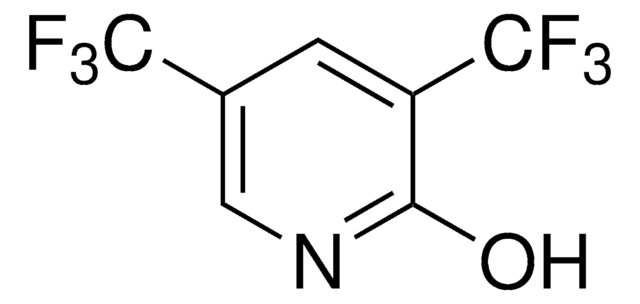
SMILES 字串
Nc1c(F)c(F)c(c(F)c1F)C(F)(F)F
InChI
1S/C7H2F7N/c8-2-1(7(12,13)14)3(9)5(11)6(15)4(2)10/h15H2
InChI 密鑰
FJOACTZFMHZHSC-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
其他說明
Used in the Preparation of
- Lactams via palladium-catalyzed olefination of arylamides with benzylacrylate, followed by 1,4-conjugate addition
- N-(fluorinated aryl)benzamides as substrates for regioselective C-H amination reactions with O-benzoylhydroxylamines
- Substituted succinimides via palladium-catalyzed carbonylation of N-aryl amides
- N-aryl cyclopropanecarboxamide substrates and various amino acid ligands for palladium-catalyzed C-H activation of cyclopropanes
相關產品
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
10 - Combustible liquids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
相关内容
The Yu program centers around the discovery of catalytic carbon–carbon and carbon–heteroatom bond forming reactions based on C–H activation. Target transformations are selected to enable 1) the use of simple and abundant starting materials such as aliphatic acids, amines and alcohols, and 2) disconnections that drastically shorten the synthesis of a drug molecule or a major class of biologically active compounds.
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门