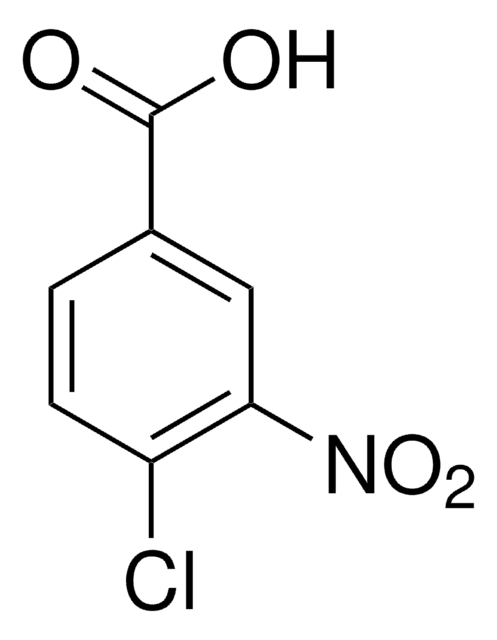
推荐产品
化驗
97%
mp
237-239 °C (lit.)
SMILES 字串
OC(=O)c1cccc(Cl)c1[N+]([O-])=O
InChI
1S/C7H4ClNO4/c8-5-3-1-2-4(7(10)11)6(5)9(12)13/h1-3H,(H,10,11)
InChI 密鑰
VCHSXYHBMFKRBK-UHFFFAOYSA-N
正在寻找类似产品? 访问 产品对比指南
一般說明
Hydrogen-bonded structures of isomeric compounds of 3-chloro-2-nitrobenzoic acid with quinoline have been investigated.
應用
3-Chloro-2-nitrobenzoic acid has been used in the preparation of:
- 2-amino-3-chlorobenzonitrile
- 3-chloro-2-nitrobenzaldehyde
訊號詞
Warning
危險聲明
危險分類
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
標靶器官
Respiratory system
儲存類別代碼
11 - Combustible Solids
水污染物質分類(WGK)
WGK 3
閃點(°F)
Not applicable
閃點(°C)
Not applicable
個人防護裝備
dust mask type N95 (US), Eyeshields, Gloves
Kazuma Gotoh et al.
Acta crystallographica. Section C, Crystal structure communications, 65(Pt 10), o534-o538 (2009-10-07)
The structures of four isomeric compounds, all C7H4ClNO4.C9H7N, of quinoline with chloro- and nitro-substituted benzoic acid, namely, 2-chloro-5-nitrobenzoic acid-quinoline (1/1), (I), 3-chloro-2-nitrobenzoic acid-quinoline (1/1), (II), 4-chloro-2-nitrobenzoic acid-quinoline (1/1), (III), and 5-chloro-2-nitrobenzoic acid-quinoline (1/1), (IV), have been determined at 185 K.
The synthesis and in vitro acetylcholinesterase and butyrylcholinesterase inhibitory activity of tacrine (Cognex?) derivaties.
Gregor VE, et al.
Bioorganic & Medicinal Chemistry Letters, 2(8), 861-864 (1992)
Tadashi Kataoka et al.
Bioorganic & medicinal chemistry, 12(9), 2397-2407 (2004-04-15)
Condensation of nitrobenzaldehydes 3 and alpha-[o-(p-methoxybenzylthio)benzoyl] sulfoxide 4 gave alpha-sulfinyl enones 5. Treatment of 5 with formic acid caused cyclization followed by debenzylation to afford 3-(methylsulfinyl)thioflavanones 6. Double-bond formation with elimination of methanesulfenic acid was performed by refluxing 6 in
我们的科学家团队拥有各种研究领域经验,包括生命科学、材料科学、化学合成、色谱、分析及许多其他领域.
联系技术服务部门