All Photos(1)
About This Item
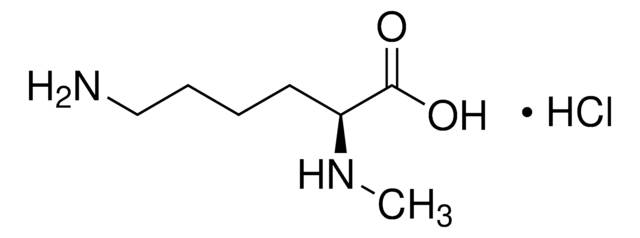
Empirical Formula (Hill Notation):
C7H16N2O2 · HCl
CAS Number:
Molecular Weight:
196.68
EC Number:
MDL number:
UNSPSC Code:
12352202
PubChem Substance ID:
NACRES:
NA.32
Assay:
≥98.0% (TLC)
Recommended Products
Quality Level
Assay
≥98.0% (TLC)
optical activity
[α]/D 20.5±1.5°, c = 0.1 in 1 M HCl
storage temp.
2-8°C
SMILES string
Cl.CNCCCC[C@H](N)C(O)=O
Cl.CNCCCC[C@H](N)C(O)=O
InChI
1S/C7H16N2O2.ClH/c1-9-5-3-2-4-6(8)7(10)11;/h6,9H,2-5,8H2,1H3,(H,10,11);1H/t6-;/m0./s1
InChI key
AQELUQTVJOFFBN-RGMNGODLSA-N
Looking for similar products? Visit Product Comparison Guide
Biochem/physiol Actions
N ε-methyl-L-lysine was identified as a lysine analog with inhibitory effects on the growth and sporulation of Penicillium chrysogenum and benzyl-penicillin formation by mycelia.
Packaging
Bottomless glass bottle. Contents are inside inserted fused cone.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
A genetically encoded epsilon-N-methyl lysine in mammalian cells.
Dan Groff et al.
Chembiochem : a European journal of chemical biology, 11(8), 1066-1068 (2010-04-28)
K Yoshioka et al.
Journal of bacteriology, 177(4), 1090-1093 (1995-02-01)
We have isolated spontaneous mutants of Salmonella typhimurium which can swim in the presence of antifilament antibodies. The molecular masses of flagellins isolated from these mutants were smaller than that (52 kDa) of wild-type flagellin. Two mutants which produced the
C G Friedrich et al.
Applied and environmental microbiology, 34(6), 706-709 (1977-12-01)
Compounds structurally related to lysine were tested against Penicillium chrysogenum Wis. 54-1255 for inhibition of growth, sporulation, and penicillin formation. This strain is relatively resistant to lysine analogs. The compounds that were the more active inhibitors of growth and whose
Y Minami et al.
Journal of biochemistry, 97(3), 745-753 (1985-03-01)
The amino acid sequence of a ferredoxin from a thermoacidophilic archaebacterium, Sulfolobus acidocaldarius, was determined by a combination of various conventional methods to be as follows: Gly-Ile-Asp-Pro-Tyr-Arg-Thr-His-Lys-Pro-Val-Val-Gly-Asp-Ser-Ser-Gly-His- Lys-Ile -Tyr-Gly-Pro-Val-Glu-Ser-Pro-Lys(Me)-Val-Leu-Gly-Val-His-Gly-Thr-Ile-Val -Gly-Va l-Asp-Phe-Asp-Leu-Cys-Ile-Ala-Asp-Gly-Ser-Cys-Ile-Thr-Ala-Cys-Pro-Val-As n-Val-P he-Gln-Trp-Tyr-Glu-Thr-Pro-Gly-His-Pro-Ala-Ser-Glu-Lys-Lys-Ala-Asp-Pro-V al-Asn- Glu-Gln-Ala-Cys-Ile-Phe-Cys-Met-Ala-Cys-Val-Asn-Val-Cys-Pro-Val-Ala-Ala- Ile-Asp -Val-Lys-Pro-Pro. It was composed
H Kalász et al.
Journal of chromatographic science, 43(4), 165-168 (2005-06-25)
Administration of (14)C-labelled L-deprenyl to rats results in the urinary elimination of a 14C-labelled compound. The 9-fluorenylmethoxycarbonyl chloride-reacted urine sample is fractionated by high-performance liquid chromatography (HPLC) on an octadecyl silica stationary phase. N(epsilon)-Monomethyl-lysine is identified in the fraction containing
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service