900878
2-Chloro-ε-caprolactone
Synonym(s):
α-Chloro-ε-caprolactone, αClεCL, 2-Chloro-1-oxacycloheptan-2-one
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C6H9ClO2
CAS Number:
Molecular Weight:
148.59
MDL number:
UNSPSC Code:
12352101
NACRES:
NA.23
Recommended Products
Assay
98% (NMR)
Quality Level
form
liquid
color
colorless to faint yellow
storage temp.
−20°C
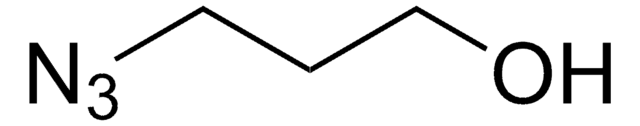
SMILES string
O=C1C(Cl)CCCCO1
General description
2-Chloro-ε-caprolactone (ClCL) is a biomaterial that can be prepared by the Baeyer-Villiger oxidation of α-chlorocyclohexanone. It can be copolymerized with ε-caprolactone for the formation of Poly(2-chloro-ε-caprolactone).
Application
2-Chloro-ε-caprolactone (or α-Chloro-ε-caprolactone) is a functionalized biodegradable monomer. This monomer can be polymerized using ring-opening polymerization to yield a chloride-functionalized polymer backbone that can either be further functionalized with small molecules or used in the synthesis of graft co-polymers.
Storage Class Code
10 - Combustible liquids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Synthesis of Poly(lactide-co-glycolide-co-ε-caprolactone)-graft-mannosylated Poly(ethylene oxide) Copolymers by Combination of ?Clip? and ?Click? Chemistries
Biomacromolecules, 13 (3), 760-78 (2012)
Development of Core?Shell Nanostructures by In Situ Assembly of Pyridine-Grafted Diblock Copolymer and Transferrin for Drug Delivery Applications
Lu L , et al.
Biomacromolecules, 17 (7), 2321-2328 (2016)
Ring-Opening Polymerization of α-Chloro-ε-caprolactone and Chemical Modification of Poly(α-chloro-ε-caprolactone
Lenoir S. et al
Macromolecules, 37 (11), 4055-4061 (2004)
Aniline-Catalyzed Reductive Amination as a Powerful Method for the Preparation of Reducing End-?Clickable? Chitooligosaccharides
Guerry A, et al.
Bioconjugate Chemistry, 24 (4), 544?9 -544?9 (2013)
Ring-opening polymerization of alpha-chloro-varepsilon-caprolactone and chemical modification of poly (alpha-chloro-varepsilon-caprolactone) by atom transfer radical processes
Lenoir S, et al.
Macromolecules, 37(11), 4055-4061 (2004)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service