740683
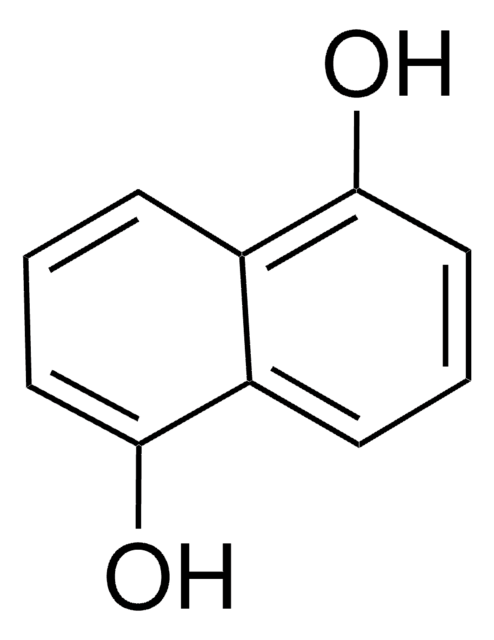
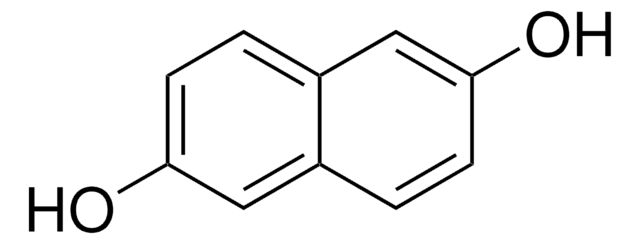
1,8-Dihydroxynaphthalene
95%
Synonym(s):
1,8-Naphthalenediol
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10H8O2
CAS Number:
Molecular Weight:
160.17
Beilstein:
2044947
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
95%
form
solid
mp
137-143 °C
storage temp.
2-8°C
SMILES string
Oc1cccc2cccc(O)c12
InChI
1S/C10H8O2/c11-8-5-1-3-7-4-2-6-9(12)10(7)8/h1-6,11-12H
InChI key
OENHRRVNRZBNNS-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Application
1,8-Dihydroxynaphthalene (DHN) can be used as:
- An intermediate in the preparation of benzo analogs of spiromamakone A.
- A starting material to synthesize naphthopyran derivatives.
- An intermediate in the total synthesis of palmarumycin CP17 analogs.
Signal Word
Danger
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Dam. 1
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Pigment biosynthesis and virulence.
A A Brakhage et al.
Contributions to microbiology, 2, 205-215 (1999-10-16)
E Thines et al.
The Journal of antibiotics, 51(4), 387-393 (1998-06-19)
From submerged cultures of Scytalidium sp. 36-93, ten metabolites were isolated due to their effects on dihydroxynaphthalene (DHN) or DOPA melanin biosynthesis. Four of the compounds, scytalols A (1a), B (1b), C (2) and D (3), are new secondary metabolites
Shao Yu Lin et al.
Molecular plant-microbe interactions : MPMI, 25(12), 1552-1561 (2012-09-01)
Both Colletotrichum and Magnaporthe spp. develop appressoria pigmented with melanin, which is essential for fungal pathogenicity. 1,8-Dihydroxynaphthalene (1,8-DHN) is believed to be polymerized to yield melanin around the appresorial cell wall through the oxidative activity of laccases. However, no 1,8-DHN
First report of a permanent open form of a naphthopyran
Martins CI, et al.
Tetrahedron Letters, 43, 2203-2205 (2002)
Total synthesis and antifungal activity of palmarumycin CP17 and its methoxy analogues
Wang R, et al.
Molecules (Basel), 21, 600-600 (2016)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service