160660
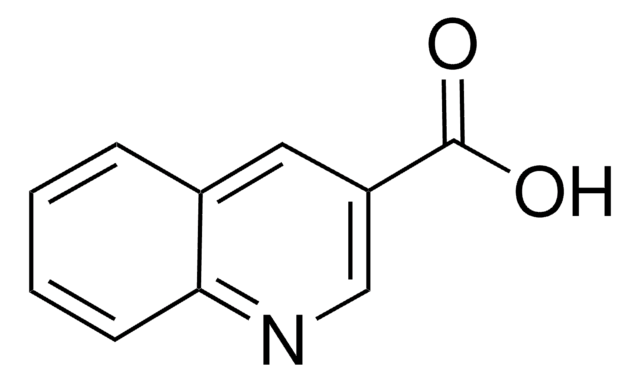
Quinaldic acid
98%
Synonym(s):
2-Quinolinecarboxylic acid
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C10H7NO2
CAS Number:
Molecular Weight:
173.17
Beilstein:
126322
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
98%
form
solid
mp
156-158 °C (lit.)
SMILES string
OC(=O)c1ccc2ccccc2n1
InChI
1S/C10H7NO2/c12-10(13)9-6-5-7-3-1-2-4-8(7)11-9/h1-6H,(H,12,13)
InChI key
LOAUVZALPPNFOQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
Quinaldic acid is also referred as quinoline-2-carboxylic acid. Microwave-assisted preparation of substituted anilides of quinaldic acid has been reported. It inhibits the oxidation of pyruvate, α-ketoglutarate, glutamate and citrate in rat liver mitochondria. Quinaldic acid is a metabolite of tryptophan degradation and inhibits the gluconeogenesis in perfused livers.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Felipe Lombó et al.
Chembiochem : a European journal of chemical biology, 7(2), 366-376 (2006-01-13)
Thiocoraline is a thiodepsipeptide antitumor compound produced by two actinomycetes Micromonospora sp. ACM2-092 and Micromonospora sp. ML1, isolated from two marine invertebrates (a soft coral and a mollusc) found of the Indian Ocean coast of Mozambique. By using oligoprimers derived
B R Bochner et al.
Journal of bacteriology, 143(2), 926-933 (1980-08-01)
A simple technique has been devised that allows direct plate selection of tetracycline-sensitive clones from a predominantly tetracycline-resistant population. The technique is especially useful in genetic methodologies based on the use of tetracycline resistance transposons, such as Tn10. Potential uses
Alleyn T Plowright et al.
Chemistry & biology, 9(5), 607-618 (2002-05-29)
Saframycin A (SafA) is a natural product that inhibits human cancer cell proliferation. Its synthetic analog, QAD, is a more potent inhibitor of these cells. SafA does not affect wild-type yeast, but it does inhibit growth of the strain CCY333
Stereocontrolled synthesis of the quinaldic acid macrocyclic system of thiostrepton.
K C Nicolaou et al.
Angewandte Chemie (International ed. in English), 41(11), 1937-1940 (2002-06-03)
Pavel Bobal et al.
Molecules (Basel, Switzerland), 17(2), 1292-1306 (2012-02-02)
In this study a one step method for the preparation of substituted anilides of quinoline-2-carboxylic acid was developed. This efficient innovative approach is based on the direct reaction of an acid or ester with substituted anilines using microwave irradiation. The
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service