242780
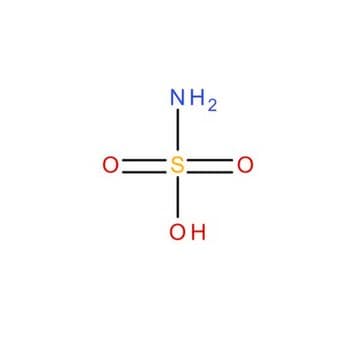
Sulfamic acid
reagent grade, 98%
Synonym(s):
Amidosulfonic acid
Sign Into View Organizational & Contract Pricing
All Photos(3)
About This Item
Linear Formula:
NH2SO3H
CAS Number:
Molecular Weight:
97.09
EC Number:
MDL number:
UNSPSC Code:
12352106
PubChem Substance ID:
NACRES:
NA.21
Recommended Products
grade
reagent grade
Quality Level
Assay
98%
form
powder or crystals
mp
215-225 °C (dec.) (lit.)
solubility
water: 213 g/L at 20 °C
water: 470 g/L at 80 °C
density
2.151 g/cm3 at 25 °C
SMILES string
NS(O)(=O)=O
InChI
1S/H3NO3S/c1-5(2,3)4/h(H3,1,2,3,4)
InChI key
IIACRCGMVDHOTQ-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
Related Categories
General description
Sulfamic acid (H2NSO3H) is a strong inorganic acid. It exists in zwitterionic form and neutral forms. Zwitterionic form is more stable than neutral form.
The ability of sulfamic acid/zinc powder to reduce nitrate to nitrogen gas under acidic condition has been utilized to denitrify nitrate-rich wastewater.
The ability of sulfamic acid/zinc powder to reduce nitrate to nitrogen gas under acidic condition has been utilized to denitrify nitrate-rich wastewater.
Application
Sulfamic acid (H2NSO3H) may be used in the following studies:
- As a catalyst in the synthesis of aryl-14H-dibenzo[a.j]xanthenes.
- As a green catalyst for the liquid Beckmann rearrangement of ketoxime in dried acetonitrile.
- As a nitrogen source as well as an in situ activator of hydroxy groups in the direct iridium-catalyzed synthesis of primary allylic amines from allylic alcohols.
- Synthesis of polysubstituted quinolones.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Aquatic Chronic 3 - Eye Irrit. 2 - Skin Irrit. 2
Storage Class Code
8A - Combustible corrosive hazardous materials
WGK
WGK 1
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
Denitrification of simulated nitrate-rich wastewater using sulfamic acid and zinc scrap.
Jang JH, et al.
Chemical Papers, 65(4), 437-446 (2011)
Sulfamic acid: a novel and efficient catalyst for the synthesis of aryl-14H-dibenzo [a. j] xanthenes under conventional heating and microwave irradiation.
Rajitha B, et al.
Tetrahedron Letters, 46(50), 8691-8693 (2005)
Solvent effects. 2. Medium effect on the structure, energy, charge density, and vibrational frequencies of sulfamic acid.
Wong MW, et al.
Journal of the American Chemical Society, 114(2), 523-529 (1992)
Sulfamic acid: an efficient, cost-effective and recyclable solid acid catalyst for the Friedlander quinoline synthesis.
Yadav JS, et al.
Tetrahedron Letters, 46(42, 7249-7253 (2005)
Sulfamic acid as a cost-effective and recyclable catalyst for liquid Beckmann rearrangement, a green process to produce amides from ketoximes without waste.
Wang B, et al.
Tetrahedron Letters, 45(17), 3369-3372 (2004)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service