188344
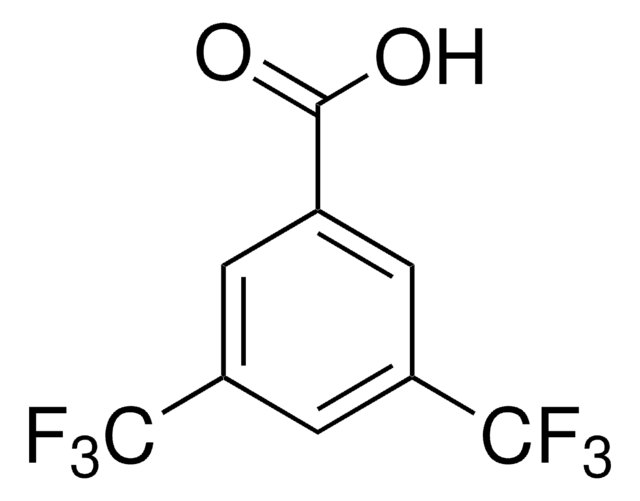
3-(Trifluoromethyl)benzoic acid
99%
Synonym(s):
α,α,α-Trifluoro-m-toluic acid, 3-Carboxybenzotrifluoride
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Linear Formula:
CF3C6H4CO2H
CAS Number:
Molecular Weight:
190.12
Beilstein:
2049239
EC Number:
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
form:
solid
Assay:
99%
Recommended Products
Quality Level
Assay
99%
form
solid
bp
238.5 °C/775 mmHg (lit.)
mp
104-106 °C (lit.)
functional group
carboxylic acid
fluoro
SMILES string
OC(=O)c1cccc(c1)C(F)(F)F
InChI
1S/C8H5F3O2/c9-8(10,11)6-3-1-2-5(4-6)7(12)13/h1-4H,(H,12,13)
InChI key
FQXQBFUUVCDIRK-UHFFFAOYSA-N
Looking for similar products? Visit Product Comparison Guide
General description
pKa values of 3-(trifluoromethyl)benzoic acid in water and in methanol has been measured. Solubility of 3-(trifluoromethyl)benzoic acid in dense carbon dioxide was evaluated to investigate the influence of fluorination on the solubility of organic pharmaceuticals in dense carbon dioxide.
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Choose from one of the most recent versions:
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
Customers Also Viewed
K H Engesser et al.
Archives of microbiology, 149(3), 188-197 (1988-01-01)
The TOL plasmid-encoded enzymes of the methylbenzoate pathway in Pseudomonas putida mt-2 cometabolized 3-trifluoromethyl (TFM)-benzoate. Two products, 3-TFM-1,2-dihydroxy-2-hydrobenzoate (3-TFM-DHB) and 2-hydroxy-6-oxo-7,7,7-trifluoro-hepta-2,4-dienoate (7-TFHOD) were identified chemically and by spectroscopic properties. TFM-substituted analogues of the metabolites of the methylbenzoate pathway were generally
S A Selifonov et al.
Biochemical and biophysical research communications, 213(3), 759-767 (1995-08-24)
Pseudomonas aeruginosa strain 142 oxidizes 2-halobenzoates via a multicomponent oxygenase (V. Romanov and R.P. Hausinger, J. Bacteriol., 1994, 176(11), 3368-3374). The intermediacy of a highly unstable cis-diol in the reaction has been proposed. Direct evidence for this is currently lacking
Alberto Gallardo et al.
Journal of biomaterials science. Polymer edition, 15(7), 917-928 (2004-08-21)
The in vitro aqueous behaviour of a metacryloyloxyethyl [2-(acetyloxy)-4-(trifluoromethyl)]benzoate (THEMA)/N,N'-dimethylacrylamide (DMA) copolymer with a THEMA molar content of 39% (labeled THDMA39) has been investigated. This composition has been selected to achieve a system able to keep both the non-water solubility
B F Taylor et al.
FEMS microbiology letters, 110(2), 213-216 (1993-06-15)
m- and p-trifluoromethyl (TFM)-benzoates are incompletely degraded by aerobic bacteria that catabolize alkylbenzoates; biodegradation ceases after ring-fission with the accumulation of a trifluoromethyl muconate semialdehyde (2-hydroxy-6-oxo-7,7,7-trifluorohepta-2,4-dienoate, TFHOD) which is resistant to biochemical attack. A bacterium (Strain V-1), isolated from sea-water
Solubility of fluorinated pharmaceuticals in dense carbon dioxide.
Laitinen A, et al.
Organic Process Research & Development, 4(5), 353-356 (2000)
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service