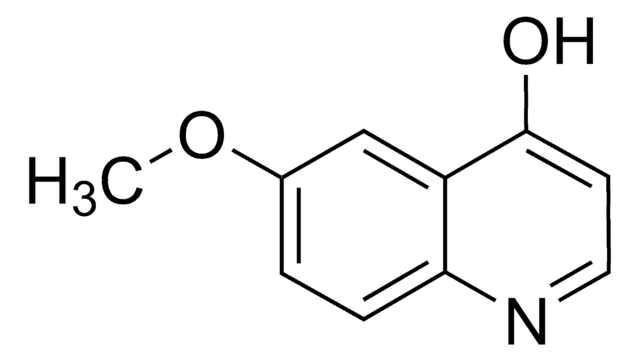
282219
Benz[g]isoquinoline-5,10-dione
99%
Synonym(s):
2-Aza-9,10-anthraquinone
Sign Into View Organizational & Contract Pricing
All Photos(1)
About This Item
Empirical Formula (Hill Notation):
C13H7NO2
CAS Number:
Molecular Weight:
209.20
MDL number:
UNSPSC Code:
12352100
PubChem Substance ID:
NACRES:
NA.22
Recommended Products
Assay
99%
mp
178-180 °C (lit.)
SMILES string
O=C1c2ccccc2C(=O)c3cnccc13
InChI
1S/C13H7NO2/c15-12-8-3-1-2-4-9(8)13(16)11-7-14-6-5-10(11)12/h1-7H
InChI key
ZLLVUAAESHIVAZ-UHFFFAOYSA-N
General description
Benz[g]isoquinoline-5,10-dione has been isolated as an active component from the ethanolic extract of the aerial parts of Mitracarpus scaber. It exhibits significant in vitro inhibitory activity against the AIDS-related pathogens. The in vitro antibacterial and anti-fungal activity of benz[g]isoquinoline-5,10-dione has been investigated using the agar well-diffusion assay.
Signal Word
Warning
Hazard Statements
Precautionary Statements
Hazard Classifications
Eye Irrit. 2 - Skin Irrit. 2 - STOT SE 3
Target Organs
Respiratory system
Storage Class Code
11 - Combustible Solids
WGK
WGK 3
Flash Point(F)
Not applicable
Flash Point(C)
Not applicable
Personal Protective Equipment
dust mask type N95 (US), Eyeshields, Gloves
Certificates of Analysis (COA)
Search for Certificates of Analysis (COA) by entering the products Lot/Batch Number. Lot and Batch Numbers can be found on a product’s label following the words ‘Lot’ or ‘Batch’.
Already Own This Product?
Find documentation for the products that you have recently purchased in the Document Library.
A M Clark et al.
Pharmaceutical research, 1(6), 269-271 (1984-11-01)
The in vitro antibacterial and anti-fungal activity of benz[g]isoquinoline-5,10-dione (1), benzo[g]quinoline-5, 10-dione (2), benzo[g]quinoline-5,6-dione (3), and anthraquinone (4) was determined using the agar well-diffusion assay. The minimum inhibitory concentrations (MIC's) of each of the active compounds (1-3) was determined using
Ekaterina Shinkevich et al.
Organic & biomolecular chemistry, 9(2), 538-548 (2010-10-27)
1,2-Disubstituted 1,2,3,4-tetrahydrobenz[g]isoquinoline-5,10-diones are prepared for the first time through an activated Pictet-Spengler reaction of the corresponding imines of 2-(1,4-dimethoxynaphth-2-yl)ethylamine in the presence of an acyl chloride and AlCl(3) followed by an oxidation with silver(II) oxide in nitric acid. Depending on
B T Walton et al.
Science (New York, N.Y.), 222(4622), 422-423 (1983-10-28)
Morphological abnormalities including extra compound eyes, extra heads, and distally duplicated legs were generated in cricket embryos by treating eggs with single doses of either benz[g]isoquinoline-5,10-dione or benzo[h]quinoline-5,6-dione. Slight structural modifications of the molecules resulted in a loss of teratogenic
Andrew Jonathan Nok
Cell biochemistry and function, 20(3), 205-212 (2002-07-19)
An ethanolic extract of Mitracarpus scaber was found to possess in vitro and in vivo trypanocidal activity against Trypanosoma congolense. At a dosage of 50 mg kg(-1) day(-1) in normal saline for 5 days, the extract cured Balbc mice infected
A L Okunade et al.
Planta medica, 65(5), 447-448 (1999-07-27)
An ethanolic extract of the aerial parts of Mitracarpus scaber demonstrated good antimicrobial activity. Bioassay directed fractionation of this extract led to the isolation of benz[g]isoquinoline-5,10-dione (1) as an active component. Compound 1 showed significant in vitro inhibitory activity against
Our team of scientists has experience in all areas of research including Life Science, Material Science, Chemical Synthesis, Chromatography, Analytical and many others.
Contact Technical Service





![1H-Benzo[g]indole 97%](/deepweb/assets/sigmaaldrich/product/structures/568/798/abc69b41-4c75-4dce-8e3a-b6ff7851c6fd/640/abc69b41-4c75-4dce-8e3a-b6ff7851c6fd.png)